Differential Roles of the Two Raphe Nuclei in Amiable Social Behavior and Aggression - An Optogenetic Study
- PMID: 30116182
- PMCID: PMC6082963
- DOI: 10.3389/fnbeh.2018.00163
Differential Roles of the Two Raphe Nuclei in Amiable Social Behavior and Aggression - An Optogenetic Study
Abstract
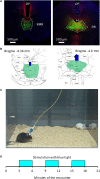
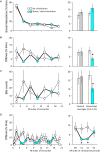
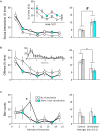
Serotonergic mechanisms hosted by raphe nuclei have important roles in affiliative and agonistic behaviors but the separate roles of the two nuclei are poorly understood. Here we studied the roles of the dorsal (DR) and median raphe region (MRR) in aggression by optogenetically stimulating the two nuclei. Mice received three 3 min-long stimulations, which were separated by non-stimulation periods of 3 min. The stimulation of the MRR decreased aggression in a phasic-like manner. Effects were rapidly expressed during stimulations, and vanished similarly fast when stimulations were halted. No carryover effects were observed in the subsequent three trials performed at 2-day intervals. No effects on social behaviors were observed. By contrast, DR stimulation rapidly and tonically promoted social behaviors: effects were present during both the stimulation and non-stimulation periods of intermittent stimulations. Aggressive behaviors were marginally diminished by acute DR stimulations, but repeated stimulations administered over 8 days considerably decreased aggression even in the absence of concurrent stimulations, indicating the emergence of carryover effects. No such effects were observed in the case of social behaviors. We also investigated stimulation-induced neurotransmitter release in the prefrontal cortex, a major site of aggression control. MRR stimulation rapidly but transiently increased serotonin release, and induced a lasting increase in glutamate levels. DR stimulation had no effect on glutamate, but elicited a lasting increase of serotonin release. Prefrontal serotonin levels remained elevated for at least 2 h subsequent to DR stimulations. The stimulation of both nuclei increased GABA release rapidly and transiently. Thus, differential behavioral effects of the two raphe nuclei were associated with differences in their neurotransmission profiles. These findings reveal a surprisingly strong behavioral task division between the two raphe nuclei, which was associated with a nucleus-specific neurotransmitter release in the prefrontal cortex.
Keywords: GABA; aggression; dorsal raphe; glutamate; median raphe; serotonin.
Figures


References
-
- Biro L., Sipos E., Bruzsik B., Farkas I., Zelena D., Balazsfi D., et al. (2018). Task division within the prefrontal cortex: distinct neuron populations selectively control different aspects of aggressive behavior via the hypothalamus. J. Neurosci. 38 4065–4075. 10.1523/JNEUROSCI.3234-17.2018 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

