IFN-γ-response mediator GBP-1 represses human cell proliferation by inhibiting the Hippo signaling transcription factor TEAD
- PMID: 30120107
- PMCID: PMC6156764
- DOI: 10.1042/BCJ20180123
IFN-γ-response mediator GBP-1 represses human cell proliferation by inhibiting the Hippo signaling transcription factor TEAD
Abstract
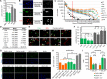
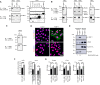
Interferon-gamma (IFN-γ) is a pleiotropic cytokine that exerts important functions in inflammation, infectious diseases, and cancer. The large GTPase human guanylate-binding protein 1 (GBP-1) is among the most strongly IFN-γ-induced cellular proteins. Previously, it has been shown that GBP-1 mediates manifold cellular responses to IFN-γ including the inhibition of proliferation, spreading, migration, and invasion and through this exerts anti-tumorigenic activity. However, the mechanisms of GBP-1 anti-tumorigenic activities remain poorly understood. Here, we elucidated the molecular mechanism of the human GBP-1-mediated suppression of proliferation by demonstrating for the first time a cross-talk between the anti-tumorigenic IFN-γ and Hippo pathways. The α9-helix of GBP-1 was found to be sufficient to inhibit proliferation. Protein-binding and molecular modeling studies revealed that the α9-helix binds to the DNA-binding domain of the Hippo signaling transcription factor TEA domain protein (TEAD) mediated by the 376VDHLFQK382 sequence at the N-terminus of the GBP-1-α9-helix. Mutation of this sequence resulted in abrogation of both TEAD interaction and suppression of proliferation. Further on, the interaction caused inhibition of TEAD transcriptional activity associated with the down-regulation of TEAD-target genes. In agreement with these results, IFN-γ treatment of the cells also impaired TEAD activity, and this effect was abrogated by siRNA-mediated inhibition of GBP-1 expression. Altogether, this demonstrated that the α9-helix is the proliferation inhibitory domain of GBP-1, which acts independent of the GTPase activity through the inhibition of the Hippo transcription factor TEAD in mediating the anti-proliferative cell response to IFN-γ.
Keywords: cell proliferation; colorectal cancer; guanylate-binding proteins; interferons.
© 2018 The Author(s).
Conflict of interest statement
The Authors declare that there are no competing interests associated with the manuscript.
Figures

Similar articles
-
Gamma interferon-induced guanylate binding protein 1 is a novel actin cytoskeleton remodeling factor.Mol Cell Biol. 2014 Jan;34(2):196-209. doi: 10.1128/MCB.00664-13. Epub 2013 Nov 4. Mol Cell Biol. 2014. PMID: 24190970 Free PMC article.
-
GBP-1 acts as a tumor suppressor in colorectal cancer cells.Carcinogenesis. 2013 Jan;34(1):153-62. doi: 10.1093/carcin/bgs310. Epub 2012 Oct 6. Carcinogenesis. 2013. PMID: 23042300
-
Pathophysiological role of guanylate-binding proteins in gastrointestinal diseases.World J Gastroenterol. 2016 Jul 28;22(28):6434-43. doi: 10.3748/wjg.v22.i28.6434. World J Gastroenterol. 2016. PMID: 27605879 Free PMC article. Review.
-
A cluster of interferon-γ-inducible p65 GTPases plays a critical role in host defense against Toxoplasma gondii.Immunity. 2012 Aug 24;37(2):302-13. doi: 10.1016/j.immuni.2012.06.009. Epub 2012 Jul 12. Immunity. 2012. PMID: 22795875
-
Regulation of the Hippo Pathway Transcription Factor TEAD.Trends Biochem Sci. 2017 Nov;42(11):862-872. doi: 10.1016/j.tibs.2017.09.003. Epub 2017 Sep 27. Trends Biochem Sci. 2017. PMID: 28964625 Free PMC article. Review.
Cited by
-
Guanylate-Binding Protein 1 as a Potential Predictor of Immunotherapy: A Pan-Cancer Analysis.Front Genet. 2022 Feb 10;13:820135. doi: 10.3389/fgene.2022.820135. eCollection 2022. Front Genet. 2022. PMID: 35222540 Free PMC article.
-
The Large GTPase, GBP-2, Regulates Rho Family GTPases to Inhibit Migration and Invadosome Formation in Breast Cancer Cells.Cancers (Basel). 2021 Nov 11;13(22):5632. doi: 10.3390/cancers13225632. Cancers (Basel). 2021. PMID: 34830789 Free PMC article.
-
Immune profiling reveals prognostic genes in high-grade serous ovarian cancer.Aging (Albany NY). 2020 Jun 16;12(12):11398-11415. doi: 10.18632/aging.103199. Epub 2020 Jun 16. Aging (Albany NY). 2020. PMID: 32544083 Free PMC article.
-
Unraveling the Role of Guanylate-Binding Proteins (GBPs) in Breast Cancer: A Comprehensive Literature Review and New Data on Prognosis in Breast Cancer Subtypes.Cancers (Basel). 2022 Jun 4;14(11):2794. doi: 10.3390/cancers14112794. Cancers (Basel). 2022. PMID: 35681772 Free PMC article.
-
Guanylate-binding protein 1 correlates with advanced tumor features, and serves as a prognostic biomarker for worse survival in lung adenocarcinoma patients.J Clin Lab Anal. 2021 Feb;35(2):e23610. doi: 10.1002/jcla.23610. Epub 2020 Dec 10. J Clin Lab Anal. 2021. PMID: 33301214 Free PMC article.
References
-
- Cheng Y.S., Colonno R.J. and Yin F.H. (1983) Interferon induction of fibroblast proteins with guanylate binding activity. J. Biol. Chem. 258, 7746–7750 PMID: - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

