Praziquantel-lipid nanocapsules: an oral nanotherapeutic with potential Schistosoma mansoni tegumental targeting
- PMID: 30122922
- PMCID: PMC6084080
- DOI: 10.2147/IJN.S167285
Praziquantel-lipid nanocapsules: an oral nanotherapeutic with potential Schistosoma mansoni tegumental targeting
Abstract
Purpose: Lipid nanocapsules (LNCs) have shown potential to increase the bioavailability and efficacy of orally administered drugs. However, their intestinal translocation to distal target sites and their implication in pharmacokinetic (PK)-pharmacodynamic (PD) relationships are yet to be elucidated. In this study, the effect of LNCs on the PD activity and pharmacokinetics of praziquantel (PZQ), the mainstay of schistosomiasis chemotherapy, was investigated.
Materials and methods: The composition of LNCs was modified to increase PZQ payload and to enhance membrane permeability. PZQ-LNCs were characterized in vitro for colloidal properties, entrapment efficiency (EE%), and drug release. PD activity of the test formulations was assessed in Schistosoma mansoni-infected mice 7 days post-oral administration of a single 250 mg/kg oral dose. Pharmacokinetics of the test formulations and their stability in simulated gastrointestinal (GI) fluids were investigated to substantiate in vivo data.
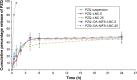
Results: PZQ-LNCs exhibited good pharmaceutical attributes in terms of size (46-62 nm), polydispersity index (0.01-0.08), EE% (>95%), and sustained release profiles. Results indicated significant efficacy enhancement by reduction in worm burden, amelioration of liver pathology, and extensive damage to the fluke suckers and tegument. This was partly explained by PK data determined in rats. In addition, oral targeting of the worms was supported by the stability of PZQ-LNCs in simulated GI fluids and scanning electron microscopy (SEM) visualization of nanostructures on the tegument of worms recovered from mesenteric/hepatic veins. Cytotoxicity data indicated tolerability of PZQ-LNCs.
Conclusion: Data obtained provide evidence for the ability of oral LNCs to target distal post-absorption sites, leading to enhanced drug efficacy. From a practical standpoint, PZQ-LNCs could be suggested as a potential tolerable single lower dose oral nanomedicine for more effective PZQ mass chemotherapy.
Keywords: Schistosoma mansoni; bioavailability; lipid nanocapsules; oral targeting; pharmacokinetics; praziquantel; tegument.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures





References
-
- Yu M, Yang Y, Zhu C, Guo S, Gan Y. Advances in the transepithelial transport of nanoparticles. Drug Discov Today. 2016;21(7):1155–1161. - PubMed
-
- Li L, Jiang G, Yu W, et al. Preparation of chitosan-based multifunctional nanocarriers overcoming multiple barriers for oral delivery of insulin. Mater Sci Eng C Mater Biol Appl. 2017;70(pt 1):278–286. - PubMed
-
- Dudhipala N, Veerabrahma K. Improved anti-hyperlipidemic activity of rosuvastatin calcium via lipid nanoparticles: pharmacokinetic and pharmacodynamic evaluation. Eur J Pharm Biopharm. 2017;110:47–57. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

