An Orphan MbtH-Like Protein Interacts with Multiple Nonribosomal Peptide Synthetases in Myxococcus xanthus DK1622
- PMID: 30126939
- PMCID: PMC6182236
- DOI: 10.1128/JB.00346-18
An Orphan MbtH-Like Protein Interacts with Multiple Nonribosomal Peptide Synthetases in Myxococcus xanthus DK1622
Abstract
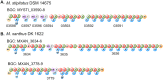
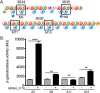
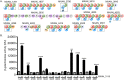
One mechanism by which bacteria and fungi produce bioactive natural products is the use of nonribosomal peptide synthetases (NRPSs). Many NRPSs in bacteria require members of the MbtH-like protein (MLP) superfamily for their solubility or function. Although MLPs are known to interact with the adenylation domains of NRPSs, the role MLPs play in NRPS enzymology has yet to be elucidated. MLPs are nearly always encoded within the biosynthetic gene clusters (BGCs) that also code for the NRPSs that interact with the MLP. Here, we identify 50 orphan MLPs from diverse bacteria. An orphan MLP is one that is encoded by a gene that is not directly adjacent to genes predicted to be involved in nonribosomal peptide biosynthesis. We targeted the orphan MLP MXAN_3118 from Myxococcus xanthus DK1622 for characterization. The M. xanthus DK1622 genome contains 15 NRPS-encoding BGCs but only one MLP-encoding gene (MXAN_3118). We tested the hypothesis that MXAN_3118 interacts with one or more NRPS using a combination of in vivo and in vitro assays. We determined that MXAN_3118 interacts with at least seven NRPSs from distinct BGCs. We show that one of these BGCs codes for NRPS enzymology that likely produces a valine-rich natural product that inhibits the clumping of M. xanthus DK1622 in liquid culture. MXAN_3118 is the first MLP to be identified that naturally interacts with multiple NRPS systems in a single organism. The finding of an MLP that naturally interacts with multiple NRPS systems suggests it may be harnessed as a "universal" MLP for generating functional hybrid NRPSs.IMPORTANCE MbtH-like proteins (MLPs) are essential accessory proteins for the function of many nonribosomal peptide synthetases (NRPSs). We identified 50 MLPs from diverse bacteria that are coded by genes that are not located near any NRPS-encoding biosynthetic gene clusters (BGCs). We define these as orphan MLPs because their NRPS partner(s) is unknown. Investigations into the orphan MLP from Myxococcus xanthus DK1622 determined that it interacts with NRPSs from at least seven distinct BGCs. Support for these MLP-NRPS interactions came from the use of a bacterial two-hybrid assay and copurification of the MLP with various NRPSs. The flexibility of this MLP to naturally interact with multiple NRPSs led us to hypothesize that this MLP may be used as a "universal" MLP during the construction of functional hybrid NRPSs.
Keywords: MbtH-like protein; Myxococcus xanthus; combinatorial biosynthesis; natural products; nonribosomal peptide synthetase.
Copyright © 2018 American Society for Microbiology.
Figures


Similar articles
-
Characterization of the Functional Variance in MbtH-like Protein Interactions with a Nonribosomal Peptide Synthetase.Biochemistry. 2017 Oct 10;56(40):5380-5390. doi: 10.1021/acs.biochem.7b00517. Epub 2017 Sep 20. Biochemistry. 2017. PMID: 28880538 Free PMC article.
-
MbtH-like proteins as integral components of bacterial nonribosomal peptide synthetases.Biochemistry. 2010 Oct 19;49(41):8815-7. doi: 10.1021/bi1012854. Biochemistry. 2010. PMID: 20845982 Free PMC article.
-
Structures of a Nonribosomal Peptide Synthetase Module Bound to MbtH-like Proteins Support a Highly Dynamic Domain Architecture.J Biol Chem. 2016 Oct 21;291(43):22559-22571. doi: 10.1074/jbc.M116.746297. Epub 2016 Sep 5. J Biol Chem. 2016. PMID: 27597544 Free PMC article.
-
[Advances in the study of the mechanism and application of nonribosomal peptide synthetases].Wei Sheng Wu Xue Bao. 2007 Aug;47(4):734-7. Wei Sheng Wu Xue Bao. 2007. PMID: 17944384 Review. Chinese.
-
Function of MbtH homologs in nonribosomal peptide biosynthesis and applications in secondary metabolite discovery.J Ind Microbiol Biotechnol. 2011 Nov;38(11):1747-60. doi: 10.1007/s10295-011-1022-8. Epub 2011 Aug 9. J Ind Microbiol Biotechnol. 2011. PMID: 21826462 Review.
Cited by
-
Leveraging synthetic biology for producing bioactive polyketides and non-ribosomal peptides in bacterial heterologous hosts.Medchemcomm. 2019 Apr 25;10(5):668-681. doi: 10.1039/c9md00055k. eCollection 2019 May 1. Medchemcomm. 2019. PMID: 31191858 Free PMC article. Review.
-
The structural basis of N-acyl-α-amino-β-lactone formation catalyzed by a nonribosomal peptide synthetase.Nat Commun. 2019 Jul 31;10(1):3432. doi: 10.1038/s41467-019-11383-7. Nat Commun. 2019. PMID: 31366889 Free PMC article.
-
Bacterial MbtH-like Proteins Stimulate Nonribosomal Peptide Synthetase-Derived Secondary Metabolism in Filamentous Fungi.ACS Synth Biol. 2019 Aug 16;8(8):1776-1787. doi: 10.1021/acssynbio.9b00106. Epub 2019 Jul 16. ACS Synth Biol. 2019. PMID: 31284717 Free PMC article.
-
TxtH is a key component of the thaxtomin biosynthetic machinery in the potato common scab pathogen Streptomyces scabies.Mol Plant Pathol. 2019 Oct;20(10):1379-1393. doi: 10.1111/mpp.12843. Epub 2019 Jul 7. Mol Plant Pathol. 2019. PMID: 31282068 Free PMC article.
-
Functional Cross-Talk of MbtH-Like Proteins During Thaxtomin Biosynthesis in the Potato Common Scab Pathogen Streptomyces scabiei.Front Microbiol. 2020 Oct 15;11:585456. doi: 10.3389/fmicb.2020.585456. eCollection 2020. Front Microbiol. 2020. PMID: 33178168 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

