B-1a cells protect mice from sepsis-induced acute lung injury
- PMID: 30134811
- PMCID: PMC6016888
- DOI: 10.1186/s10020-018-0029-2
B-1a cells protect mice from sepsis-induced acute lung injury
Abstract
Background: Sepsis morbidity and mortality are aggravated by acute lung injury (ALI) or acute respiratory distress syndrome (ARDS). Mouse B-1a cells are a phenotypically and functionally unique sub-population of B cells, providing immediate protection against infection by releasing natural antibodies and immunomodulatory molecules. We hypothesize that B-1a cells ameliorate sepsis-induced ALI.
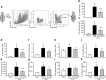
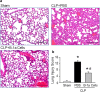
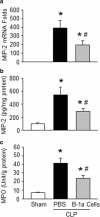
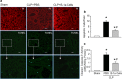
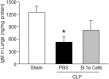
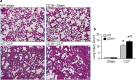
Methods: Sepsis was induced in C57BL/6 mice by cecal ligation and puncture (CLP). PBS or B-1a cells were adoptively transferred into the septic mice intraperitoneally. After 20 h of CLP, lungs were harvested and assessed by PCR and ELISA for pro-inflammatory cytokines (IL-6, IL-1β) and chemokine (MIP-2) expression, by histology for injury, by TUNEL and cleaved caspase-3 for apoptosis, and by myeloperoxidase (MPO) assay for neutrophil infiltration.
Results: We found that septic mice adoptively transferred with B-1a cells significantly decreased the mRNA and protein levels of IL-6, IL-1β and MIP-2 in the lungs compared to PBS-treated mice. Mice treated with B-1a cells showed dramatic improvement in lung injury compared to PBS-treated mice after sepsis. We found apoptosis in the lungs was significantly inhibited in B-1a cell injected mice compared to PBS-treated mice after sepsis. B-1a cell treatment significantly down-regulated MPO levels in the lungs compared to PBS-treated mice in sepsis. The protective outcomes of B-1a cells in ALI was further confirmed by using B-1a cell deficient CD19-/- mice, which showed significant increase in the lung injury scores following sepsis as compared to WT mice.
Conclusions: Our results demonstrate a novel therapeutic potential of B-1a cells to treat sepsis-induced ALI.
Keywords: Acute lung injury; B-1a cells; IL-10; Inflammation; Neutrophils; Sepsis.
Conflict of interest statement
Ethics approval
All animal protocols were approved by our Institutional Animal Care and Use Committee of the Feinstein Institute for Medical Research.
Consent for publication
All authors have contributed to, read and approved the final version of this manuscript for submission and publication in the journal
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

