An extracellular matrix fragment drives epithelial remodeling and airway hyperresponsiveness
- PMID: 30135247
- PMCID: PMC6342449
- DOI: 10.1126/scitranslmed.aaq0693
An extracellular matrix fragment drives epithelial remodeling and airway hyperresponsiveness
Abstract
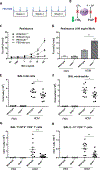
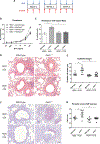
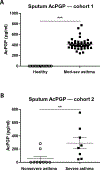
It is anticipated that bioactive fragments of the extracellular matrix (matrikines) can influence the development and progression of chronic diseases. The enzyme leukotriene A4 hydrolase (LTA4H) mediates opposing proinflammatory and anti-inflammatory activities, through the generation of leukotriene B4 (LTB4) and degradation of proneutrophilic matrikine Pro-Gly-Pro (PGP), respectively. We show that abrogation of LTB4 signaling ameliorated inflammation and airway hyperresponsiveness (AHR) in a murine asthma model, yet global loss of LTA4H exacerbated AHR, despite the absence of LTB4 This exacerbated AHR was attributable to a neutrophil-independent capacity of PGP to promote pathological airway epithelial remodeling. Thus, we demonstrate a disconnect between airway inflammation and AHR and the ability of a matrikine to promote an epithelial remodeling phenotype that negatively affects lung function. Subsequently, we show that substantial quantities of PGP are detectable in the sputum of moderate-severe asthmatics in two distinct cohorts of patients. These studies have implications for our understanding of remodeling phenotypes in asthma and may rationalize the failure of LTA4H inhibitors in the clinic.
Copyright © 2018 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures




Comment in
-
Response to Comment on "An extracellular matrix fragment drives epithelial remodeling and airway hyperresponsiveness".Sci Transl Med. 2019 Jun 19;11(497):eaaw0462. doi: 10.1126/scitranslmed.aaw0462. Sci Transl Med. 2019. PMID: 31217333
-
Comment on "An extracellular matrix fragment drives epithelial remodeling and airway hyperresponsiveness".Sci Transl Med. 2019 Jun 19;11(497):eaav4538. doi: 10.1126/scitranslmed.aav4538. Sci Transl Med. 2019. PMID: 31217337
References
-
- Saglani S, Lloyd CM, Novel concepts in airway inflammation and remodelling in asthma. Eur. Respir. J 46, 1796–1804 (2015). - PubMed
-
- Haeggström JZ, Tholander F, Wetterholm A, Structure and catalytic mechanisms of leukotriene A4 hydrolase. Prostaglandins Other Lipid Mediat. 83, 198–202 (2007). - PubMed
-
- Tager AM, Luster AD, BLT1 and BLT2: The leukotriene B4 receptors. Prostaglandins Leukot. Essent. Fatty Acids 69, 123–134 (2003). - PubMed
-
- Di Gennaro A, Haeggström JZ, The leukotrienes: Immune-modulating lipid mediators of disease. Adv. Immunol 116, 51–92 (2012). - PubMed
-
- Di Gennaro A, Haeggström JZ, Targeting leukotriene B4 in inflammation. Expert Opin. Ther. Targets 18, 79–93 (2014). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL110950/HL/NHLBI NIH HHS/United States
- 209458/Z/17/Z/WT_/Wellcome Trust/United Kingdom
- I01 BX001756/BX/BLRD VA/United States
- R01 HL126596/HL/NHLBI NIH HHS/United States
- MC_PC_13064/MRC_/Medical Research Council/United Kingdom
- 202865/Z/16/Z/WT_/Wellcome Trust/United Kingdom
- R01 HL077783/HL/NHLBI NIH HHS/United States
- R01 HL136211/HL/NHLBI NIH HHS/United States
- R01 HL114439/HL/NHLBI NIH HHS/United States
- 107059/Z/15/Z/WT_/Wellcome Trust/United Kingdom
- G1000758/MRC_/Medical Research Council/United Kingdom
- R01 HL102371/HL/NHLBI NIH HHS/United States
- R01 HL122426/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

