Hypocretin Mediates Sleep and Wake Disturbances in a Mouse Model of Traumatic Brain Injury
- PMID: 30136622
- PMCID: PMC6387567
- DOI: 10.1089/neu.2018.5810
Hypocretin Mediates Sleep and Wake Disturbances in a Mouse Model of Traumatic Brain Injury
Abstract
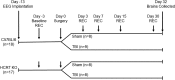
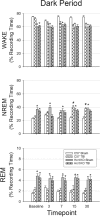
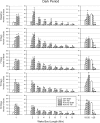
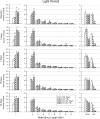
Traumatic brain injury (TBI) is a major cause of disability worldwide. Post-TBI sleep and wake disturbances are extremely common and difficult for patients to manage. Sleep and wake disturbances contribute to poor functional and emotional outcomes from TBI, yet effective therapies remain elusive. A more comprehensive understanding of mechanisms underlying post-TBI sleep and wake disturbance will facilitate development of effective pharmacotherapies. Previous research in human patients and animal models indicates that altered hypocretinergic function may be a major contributor to sleep-wake disturbance after TBI. In this study, we further elucidate the role of hypocretin by determining the impact of TBI on sleep-wake behavior of hypocretin knockout (HCRT KO) mice. Adult male C57BL/6J and HCRT KO mice were implanted with electroencephalography recording electrodes, and pre-injury baseline recordings were obtained. Mice were then subjected to either moderate TBI or sham surgery. Additional recordings were obtained and sleep-wake behavior determined at 3, 7, 15, and 30 days after TBI or sham procedures. At baseline, HCRT KO mice had a significantly different sleep-wake phenotype than control C57BL/6J mice. Post-TBI sleep-wake behavior was altered in a genotype-dependent manner: sleep of HCRT KO mice was not altered by TBI, whereas C57BL/6J mice had more non-rapid eye movement sleep, less wakefulness, and more short wake bouts and fewer long wake bouts. Numbers of hypocretin-positive cells were reduced in C57BL/6J mice by TBI. Collectively, these data indicate that the hypocretinergic system is involved in the alterations in sleep-wake behavior that develop after TBI in this model, and suggest potential therapeutic interventions.
Keywords: TBI; brain injury; controlled cortical impact; orexin; sleep; wake.
Conflict of interest statement
No competing financial interests exist.
Figures


References
-
- Thurman D., Alverson C., and Dunn K. (1999). Traumatic brain injury in the United States: a public health perspective. J. Head Trauma Rehabil. 14, 602–615 - PubMed
-
- Ma V.Y., Chan L., and Carruthers K.J. (2014). Incidence, prevalence, costs, and impact on disability of common conditions requiring rehabilitation in the United States: stroke, spinal cord injury, traumatic brain injury, meultiple sclerosis, osteoarthritis, rheumatoid arthritis, limb loss, and back pain. Arch. Phys. Med. Rehabil. 95, 986–995 .e1 - PMC - PubMed
-
- Baumann C.R. (2016). Sleep and traumatic brain injury. Sleep Med. Clin. 11, 19–23 - PubMed
-
- Wiseman-Hakes C., Duclos C., Blais H., Dumont M., Bernard F., Desautels A., Menon D., Gilbert D. and Carrier J. (2016). Sleep in the acute phase of severe traumatic brain injury: a snapshot of polysomnography. Neurorehabil. Neural Repair 30, 713–721 - PubMed
-
- Chiu H.Y., Chen P.Y., Chen N.H., Chuang L.P., and Tsai P.S. (2013). Trajectories of sleep changes during the acute phase of traumatic brain injury: a 7-day actigraphy study. J. Formos. Med. Assoc. 112, 545–553 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

