Can neuroimaging predict dementia in Parkinson's disease?
- PMID: 30137209
- PMCID: PMC6113860
- DOI: 10.1093/brain/awy211
Can neuroimaging predict dementia in Parkinson's disease?
Abstract
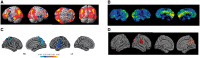
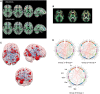
Dementia in Parkinson's disease affects 50% of patients within 10 years of diagnosis but there is wide variation in severity and timing. Thus, robust neuroimaging prediction of cognitive involvement in Parkinson's disease is important: (i) to identify at-risk individuals for clinical trials of potential new treatments; (ii) to provide reliable prognostic information for individuals and populations; and (iii) to shed light on the pathophysiological processes underpinning Parkinson's disease dementia. To date, neuroimaging has not made major contributions to predicting cognitive involvement in Parkinson's disease. This is perhaps unsurprising considering conventional methods rely on macroscopic measures of topographically distributed neurodegeneration, a relatively late event in Parkinson's dementia. However, new technologies are now emerging that could provide important insights through detection of other potentially relevant processes. For example, novel MRI approaches can quantify magnetic susceptibility as a surrogate for tissue iron content, and increasingly powerful mathematical approaches can characterize the topology of brain networks at the systems level. Here, we present an up-to-date overview of the growing role of neuroimaging in predicting dementia in Parkinson's disease. We discuss the most relevant findings to date, and consider the potential of emerging technologies to detect the earliest signs of cognitive involvement in Parkinson's disease.
Figures
References
-
- Aarsland D, Brønnick K, Larsen JP, Tysnes OB, Alves G. Cognitive impairment in incident, untreated parkinson disease: the norwegian parkwest study. Neurology 2009; 72: 1121–26. - PubMed
-
- Acosta-Cabronero J, Cardenas-Blanco A, Betts MJ, Butryn M, Valdes-Herrera JP, Galazky I et al. . The whole-brain pattern of magnetic susceptibility perturbations in Parkinson’s disease. Brain 2016; 140: 118–31. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

