Short-term effects of low-concentration atropine eye drops on pupil size and accommodation in young adult subjects
- PMID: 30145612
- PMCID: PMC6208716
- DOI: 10.1007/s00417-018-4112-8
Short-term effects of low-concentration atropine eye drops on pupil size and accommodation in young adult subjects
Abstract
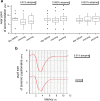
Purpose: A single eye drop containing 0.01% atropine every evening has previously been found to inhibit myopia progression in young adults. We have tested the short-term effects of very low-dose atropine eye drops on pupil sizes and accommodation in young adult subjects.
Methods: Fourteen eyes of young adult subjects participated in the clinical observation. A single eye drop was applied with concentrations of either 0.01%, 0.005%, or 0.001% in the evening. Baseline parameters were measured before atropine application. Changes of pupil sizes, under photopic and mesopic conditions, as well as accommodation amplitudes were observed over the next day and analyzed by paired the Wilcoxon signed-rank test.
Results: The pupil was significantly dilated 12 h after instillation of 0.01% atropine eye drops, both under photopic (3.3 ± 0.5 mm vs. 4.9 ± 0.9 mm) and mesopic (4.8 ± 0.7 mm vs. 6.1 ± 0.7 mm) conditions. Pupil sizes recovered over the day but were still significantly larger in the evening, compared to the baseline parameters measured on the day before (3.9 ± 0.5 mm vs. 5.3 ± 0.6 mm). The subjective near point of accommodation was reduced from 8.0 ± 2.4 to 6.6 ± 2.8 dpt in the morning and to 7.0 ± 2.9 dpt in the evening. At 0.005%, the pattern of results remained still similar, although the magnitude of the effects was generally smaller. At 0.001%, pupil sizes were still weakly significantly larger in the morning.
Conclusions: At a dose of 0.01%, clinically significant short-term effects were detected on pupil size and accommodation for at least 24 h. At the lowest dose of 0.001%, only tiny effects on pupil size were detectable.
Keywords: Accommodation; Atropine; Eye drops; Myopia; Pupil size.
Conflict of interest statement
Conflict of interest
Author A Fricke has received a speaker honorarium of DGII in 2016.
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest other than the speaker honorarium (A Fricke) (such as honoraria; educational grants; participation in speaker bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements) or non-financial interest (such as personal or professional relationship, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

