Mouse Motor Cortex Coordinates the Behavioral Response to Unpredicted Sensory Feedback
- PMID: 30146302
- PMCID: PMC6127035
- DOI: 10.1016/j.neuron.2018.07.046
Mouse Motor Cortex Coordinates the Behavioral Response to Unpredicted Sensory Feedback
Erratum in
-
Mouse Motor Cortex Coordinates the Behavioral Response to Unpredicted Sensory Feedback.Neuron. 2019 Mar 20;101(6):1202. doi: 10.1016/j.neuron.2019.02.042. Neuron. 2019. PMID: 30897361 Free PMC article. No abstract available.
Abstract
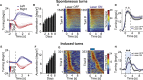
Motor cortex (M1) lesions result in motor impairments, yet how M1 contributes to the control of movement remains controversial. To investigate the role of M1 in sensory guided motor coordination, we trained mice to navigate a virtual corridor using a spherical treadmill. This task required directional adjustments through spontaneous turning, while unexpected visual offset perturbations prompted induced turning. We found that M1 is essential for execution and learning of this visually guided task. Turn-selective layer 2/3 and layer 5 pyramidal tract (PT) neuron activation was shaped differentially with learning but scaled linearly with turn acceleration during spontaneous turns. During induced turns, however, layer 2/3 neurons were activated independent of behavioral response, while PT neurons still encoded behavioral response magnitude. Our results are consistent with a role of M1 in the detection of sensory perturbations that result in deviations from intended motor state and the initiation of an appropriate corrective response.
Copyright © 2018 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures







Comment in
-
Circuits for Raiders.Neuron. 2018 Sep 5;99(5):872-873. doi: 10.1016/j.neuron.2018.08.024. Neuron. 2018. PMID: 30189206
References
-
- Attinger A., Wang B., Keller G.B. Visuomotor Coupling Shapes the Functional Development of Mouse Visual Cortex. Cell. 2017;169:1291–1302.e14. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

