MBD2 regulates differentiation and function of Th17 cells in neutrophils- dominant asthma via HIF-1α
- PMID: 30150897
- PMCID: PMC6102869
- DOI: 10.1186/s12950-018-0191-x
MBD2 regulates differentiation and function of Th17 cells in neutrophils- dominant asthma via HIF-1α
Abstract
Background: T helper 17 (Th17) cells have proven to be crucial in the pathogenesis of neutrophils-dominant asthma. Hypoxia inducible factor-1α (HIF-1α) is involved in allergic responses in asthma. Our previous studies indicated that Methtyl-CpG binding domain protein 2 (MBD2) expression was increased in asthma patients. The aim of the present study is to understand how MBD2 interacts with HIF-1α to regulate Th17 cell differentiation and IL-17 expression in neutrophils-dominant asthma.
Methods: A neutrophils-dominant asthma mouse model was established using female C57BL/6 mice to investigate Th17 cell differentiation and MBD2 and HIF-1α expression regulation using flow cytometry, western blot or qRT-PCR. MBD2 and HIF-1α genes were silenced or overexpressed through lentiviral transduction to explore the roles of MBD2 in Th17 cell differentiation and IL-17 release in neutrophils-dominant asthma.
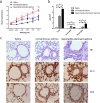
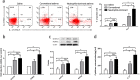
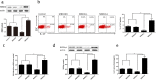
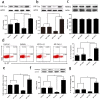
Results: A neutrophilic inflammatory asthma phenotype model was established successfully. This was characterized by airway hyperresponsiveness (AHR), increased BALF neutrophil granulocytes, activated Th17 cell differentiation, and high IL-17 levels. MBD2 and HIF-1α expression were significantly increased in the lung and spleen cells of mice with neutrophils-dominant asthma. Through overexpression or silencing of MBD2 and HIF-1α genes, we have concluded that MBD2 and HIF-1α regulate Th17 cell differentiation and IL-17 secretion. Moreover, MBD2 was also found to regulate HIF-1α expression.
Conclusions: Our findings have uncovered new roles for MBD2 and HIF-1α, and provide novel insights into the epigenetic regulation of neutrophils-dominant asthma.
Keywords: Hypoxia inducible factor-1α; Methtyl-CpG binding domain protein 2; Neutrophils-dominant asthma; T helper 17 cells.
Conflict of interest statement
Ethical approval, all applicable international, national, and/or institutional guidelines for the care and use of animals were followed.Not applicable.The authors declare that they have no competing interests.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures



Similar articles
-
Role of Sex Hormones at Different Physiobiological Conditions and Therapeutic Potential in MBD2 Mediated Severe Asthma.Oxid Med Cell Longev. 2021 Dec 14;2021:7097797. doi: 10.1155/2021/7097797. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 35096261 Free PMC article. Review.
-
MBD2 mediates Th17 cell differentiation by regulating MINK1 in Th17-dominant asthma.Front Genet. 2022 Oct 11;13:959059. doi: 10.3389/fgene.2022.959059. eCollection 2022. Front Genet. 2022. PMID: 36303542 Free PMC article.
-
MBD2-mediated Th17 differentiation in severe asthma is associated with impaired SOCS3 expression.Exp Cell Res. 2018 Oct 1;371(1):196-204. doi: 10.1016/j.yexcr.2018.08.010. Epub 2018 Aug 9. Exp Cell Res. 2018. PMID: 30098334
-
miR-146a-3p as a potential novel therapeutic by targeting MBD2 to mediate Th17 differentiation in Th17 predominant neutrophilic severe asthma.Clin Exp Med. 2023 Oct;23(6):2839-2854. doi: 10.1007/s10238-023-01033-0. Epub 2023 Mar 24. Clin Exp Med. 2023. PMID: 36961677 Free PMC article.
-
Neutrophilic inflammation in severe asthma.Int Arch Allergy Immunol. 2012;158 Suppl 1:96-102. doi: 10.1159/000337801. Epub 2012 May 15. Int Arch Allergy Immunol. 2012. PMID: 22627375 Review.
Cited by
-
Resolution of allergic asthma.Semin Immunopathol. 2019 Nov;41(6):665-674. doi: 10.1007/s00281-019-00770-3. Epub 2019 Nov 8. Semin Immunopathol. 2019. PMID: 31705318 Review.
-
Role of Sex Hormones at Different Physiobiological Conditions and Therapeutic Potential in MBD2 Mediated Severe Asthma.Oxid Med Cell Longev. 2021 Dec 14;2021:7097797. doi: 10.1155/2021/7097797. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 35096261 Free PMC article. Review.
-
The methyl-CpG binding domain 2 regulates peptidylarginine deiminase 4 expression and promotes neutrophil extracellular trap formation via the Janus kinase 2 signaling pathway in experimental severe asthma.Ann Med. 2025 Dec;57(1):2458207. doi: 10.1080/07853890.2025.2458207. Epub 2025 Jan 27. Ann Med. 2025. PMID: 39865866 Free PMC article.
-
MBD2 mediates Th17 cell differentiation by regulating MINK1 in Th17-dominant asthma.Front Genet. 2022 Oct 11;13:959059. doi: 10.3389/fgene.2022.959059. eCollection 2022. Front Genet. 2022. PMID: 36303542 Free PMC article.
-
Increased HERV-E clone 4-1 expression contributes to DNA hypomethylation and IL-17 release from CD4+ T cells via miR-302d/MBD2 in systemic lupus erythematosus.Cell Commun Signal. 2019 Aug 14;17(1):94. doi: 10.1186/s12964-019-0416-5. Cell Commun Signal. 2019. PMID: 31412880 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

