Microbial larvicides for mosquito control: Impact of long lasting formulations of Bacillus thuringiensis var. israelensis and Bacillus sphaericus on non-target organisms in western Kenya highlands
- PMID: 30151171
- PMCID: PMC6106196
- DOI: 10.1002/ece3.4250
Microbial larvicides for mosquito control: Impact of long lasting formulations of Bacillus thuringiensis var. israelensis and Bacillus sphaericus on non-target organisms in western Kenya highlands
Abstract
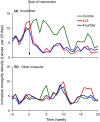
The microbial larvicides Bacillus thuringiensis var. israelensis and Bacillus sphaericus have been used extensively for mosquito control and have been found to be effective and safe to non-target organisms cohabiting with mosquito larvae. Recently developed long lasting microbial larvicides (LLML), although evading the previous challenge of short duration of activity, increase the risk of persistence of toxins in the treated larval habitats. This study monitored the impact of LLML FourStar® and LL3 on non-target organisms cohabiting with mosquito larvae in an operational study to control malaria vectors in western Kenya highlands. A total of 300 larval habitats were selected in three highland villages. The habitats were first monitored for 5 weeks to collect baseline data on non-target organisms cohabiting with mosquito larvae and then randomized into two treatment arms (respective FourStar® and LL3) and one control arm. Non-target organisms were sampled weekly for 5 months after treatment to assess the impact of LLML intervention. Before treatment, the mean density of all non-target organisms combined in the control, LL3 and FourStar® treated habitats was 1.42, 1.39 and 1.49 individuals per habitat per sampling occasion, respectively. Following treatment, this density remained fairly unchanged for 21 weeks at which time it was 1.82, 2.11, and 2.05 for the respective control, LL3 and FourStar® treated habitats. Statistical analysis revealed that LL3 and FourStar® did not significantly alter abundance, richness or diversity of the 11 taxa studied, when comparing the intervention and control larval habitats. However, both FourStar® and LL3 significantly reduced the density of malaria vectors. In conclusion, one round of label rate application of FourStar® or LL3 in natural larval habitats did not alter richness, abundance or diversity of the monitored aquatic non-target organisms cohabiting with mosquito larvae to an ecologically significant level.
Keywords: Bacillus sphaericus; Bacillus thuringiensis var. israelensis; aquatic vertebrates and invertebrates; larviciding.
Figures


References
-
- Åbjörnsson, K. , Brönmark, C. , & Hansson, L. A. (2002). The relative importance of lethal and non‐lethal effects of fish on insect colonisation of ponds. Freshwater Biology, 47, 1489–1495. 10.1046/j.1365-2427.2002.00883.x - DOI
-
- Boisvert, M. , & Boisvert, J. (1999). Persistence of toxic activity and recycling of Bacillus thuringiensis var. israelensis in cold water: Field experiments using diffusion chambers in a pond. Biocontrol Science and Technology, 9(4), 507–522. 10.1080/09583159929479 - DOI
-
- Boisvert, M. , & Boisvert, J. (2000). Effects of Bacillus thuringiensis var. israelensis on target and non target organisms: A review of laboratory and field experiments. Biocontrol Science and Technology, 10, 517–561. 10.1080/095831500750016361 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

