Using hyperpolarised NMR and DFT to rationalise the unexpected hydrogenation of quinazoline to 3,4-dihydroquinazoline
- PMID: 30152480
- PMCID: PMC6136267
- DOI: 10.1039/c8cc04826f
Using hyperpolarised NMR and DFT to rationalise the unexpected hydrogenation of quinazoline to 3,4-dihydroquinazoline
Abstract
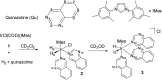
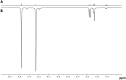
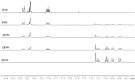
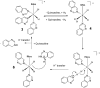
PHIP and SABRE hyperpolarized NMR methods are used to follow the unexpected metal-catalysed hydrogenation of quinazoline (Qu) to 3,4-dihydroquinazoline as the sole product. A solution of [IrCl(IMes)(COD)] in dichloromethane reacts with H2 and Qu to form [IrCl(H)2(IMes)(Qu)2] (2). The addition of methanol then results in its conversion to [Ir(H)2(IMes)(Qu)3]Cl (3) which catalyses the hydrogenation reaction. Density functional theory calculations are used to rationalise a proposed outer sphere mechanism in which (3) converts to [IrCl(H)2(H2)(IMes)(Qu)2]Cl (4) and neutral [Ir(H)3(IMes)(Qu)2] (6), both of which are involved in the formation of 3,4-dihydroquinazoline via the stepwise transfer of H+ and H-, with H2 identified as the reductant. Successive ligand exchange in 3 results in the production of thermodynamically stable [Ir(H)2(IMes)(3,4-dihydroquinazoline)3]Cl (5).
Figures
Similar articles
-
Reaction Monitoring Using SABRE-Hyperpolarized Benchtop (1 T) NMR Spectroscopy.Anal Chem. 2019 May 21;91(10):6695-6701. doi: 10.1021/acs.analchem.9b00729. Epub 2019 May 2. Anal Chem. 2019. PMID: 30985110 Free PMC article.
-
Investigating pyridazine and phthalazine exchange in a series of iridium complexes in order to define their role in the catalytic transfer of magnetisation from para-hydrogen.Chem Sci. 2015 Jul 1;6(7):3981-3993. doi: 10.1039/c5sc00756a. Epub 2015 Apr 28. Chem Sci. 2015. PMID: 29218168 Free PMC article.
-
Iridium trihydride and tetrahydride complexes and their role in catalytic polarisation transfer from parahydrogen to pyruvate.Chem Sci. 2024 Dec 12;16(3):1396-1404. doi: 10.1039/d4sc06138a. eCollection 2025 Jan 15. Chem Sci. 2024. PMID: 39703413 Free PMC article.
-
Iridium(III) hydrido N-heterocyclic carbene-phosphine complexes as catalysts in magnetization transfer reactions.Inorg Chem. 2013 Dec 2;52(23):13453-61. doi: 10.1021/ic401783c. Epub 2013 Nov 11. Inorg Chem. 2013. PMID: 24215616 Free PMC article.
-
Advancing homogeneous catalysis for parahydrogen-derived hyperpolarisation and its NMR applications.Chem Sci. 2022 Mar 22;13(17):4670-4696. doi: 10.1039/d2sc00737a. eCollection 2022 May 4. Chem Sci. 2022. PMID: 35655870 Free PMC article. Review.
Cited by
-
Signal amplification by reversible exchange for COVID-19 antiviral drug candidates.Sci Rep. 2020 Aug 31;10(1):14290. doi: 10.1038/s41598-020-71282-6. Sci Rep. 2020. PMID: 32868801 Free PMC article.
-
Electrophilically Activated Nitroalkanes in Synthesis of 3,4-Dihydroquinozalines.Molecules. 2021 Jul 14;26(14):4274. doi: 10.3390/molecules26144274. Molecules. 2021. PMID: 34299549 Free PMC article.
-
Hyperpolarization of Pyridyl Fentalogues by Signal Amplification By Reversible Exchange (SABRE).ChemistryOpen. 2019 Nov 8;8(12):1375-1382. doi: 10.1002/open.201900273. eCollection 2019 Dec. ChemistryOpen. 2019. PMID: 31844604 Free PMC article.
-
Deactivation of catalysts in simultaneous reversible and irreversible parahydrogen NMR signal enhancement, and the role of co-ligands in the stabilization of the reversible method.RSC Adv. 2022 May 27;12(25):15986-15991. doi: 10.1039/d2ra02872g. eCollection 2022 May 23. RSC Adv. 2022. PMID: 35733673 Free PMC article.
References
-
- Guenther U. L., Modern NMR Methodology, 2013, vol. 335, pp. 23–69. - PubMed
-
- Oros A. M., Shah N. J. Phys. Med. Biol. 2004;49:R105–R153. - PubMed
-
- Viale A., Reineri F., Santelia D., Cerutti E., Ellena S., Gobetto R., Aime S. Q. J. Nucl. Med. Mol. Imaging. 2009;53:604–617. - PubMed
-
- Lee D., Monin G., Duong N. T., Lopez I. Z., Bardet M., Mareau V., Gonon L., De Paepe G. J. Am. Chem. Soc. 2014;136:13781–13788. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

