Zoledronic acid in the management of mesothelioma - a feasibility study (Zol-A Trial): study protocol for a randomised controlled trial
- PMID: 30157910
- PMCID: PMC6116562
- DOI: 10.1186/s13063-018-2851-9
Zoledronic acid in the management of mesothelioma - a feasibility study (Zol-A Trial): study protocol for a randomised controlled trial
Abstract
Background: Nitrogen containing bisphosphonates such as zoledronic acid (ZA) are known to contain certain anti-cancer properties. These have been investigated in the past in various cancers such as breast, prostate and colon. ZA in particular has shown promising results in pre-clinical studies. We propose a multicentre double-blind randomised controlled feasibility study to assess the recruitment and acceptability of ZA/placebo alongside chemotherapy in malignant pleural mesothelioma (MPM).
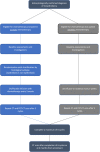
Methods: Patients will be recruited for a 13-month period from October 2016 to November 2017. Eligible patients will be identified via the regional mesothelioma multidisciplinary team meeting. Those who receive chemotherapy will be randomised to receive either ZA or placebo alongside their chemotherapy. Those who decline chemotherapy will be offered to join the trial on the non-randomised open-labelled arm of the trial. Patients will receive a maximum of six cycles of ZA/placebo, at three-weekly cycles. All patients will be followed up for six months from randomisation. Semi-structured interviews to gather data on acceptability of trial procedures, tolerability of ZA and other relevant information will take place after the participants have completed their six cycles of treatment. For a better understanding about non-participation in mesothelioma trials we also aim to interview those who decline to take part in the trial.
Discussion: The qualitative and quantitative data gathered in this feasibility trial will hopefully pave the way to designing a robust full phase III trial to investigate the potential synergistic effect of ZA and current standard treatment for MPM, cisplatin-pemetrexed combination chemotherapy.
Trial registration: ISRCTN Registry, ISRCTN45536692 . Registered on 9 August 2016. EudraCT no. 2015-004433-26.
Keywords: Malignant pleural mesothelioma; Mesothelin; Mesothelioma; PET-CT; Zoledronic acid.
Conflict of interest statement
Ethics approval and consent to participate
The study protocol and related documents were reviewed by East of England Cambridge East Research Ethics Committee (Ref 16/EE/0105). All participants will be asked for written consent before enrolling in the study. Participants will be given sufficient time to read the PIL and ask any questions relating to the study before consenting. Participants will also have the right to withdraw from the study at any time without having to give reason, if they change their mind regarding participation.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Durvalumab with first-line chemotherapy in previously untreated malignant pleural mesothelioma (DREAM): a multicentre, single-arm, phase 2 trial with a safety run-in.Lancet Oncol. 2020 Sep;21(9):1213-1223. doi: 10.1016/S1470-2045(20)30462-9. Lancet Oncol. 2020. PMID: 32888453 Clinical Trial.
-
Nintedanib in combination with pemetrexed and cisplatin for chemotherapy-naive patients with advanced malignant pleural mesothelioma (LUME-Meso): a double-blind, randomised, placebo-controlled phase 3 trial.Lancet Respir Med. 2019 Jul;7(7):569-580. doi: 10.1016/S2213-2600(19)30139-0. Epub 2019 May 15. Lancet Respir Med. 2019. PMID: 31103412 Clinical Trial.
-
Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial.Lancet. 2016 Apr 2;387(10026):1405-1414. doi: 10.1016/S0140-6736(15)01238-6. Epub 2015 Dec 21. Lancet. 2016. PMID: 26719230 Clinical Trial.
-
[Chemotherapy of malignant pleural mesothelioma: have we made any progress?].Zentralbl Chir. 2008 Jun;133(3):238-42. doi: 10.1055/s-2008-1004809. Zentralbl Chir. 2008. PMID: 18563688 Review. German.
-
Pemetrexed-cisplatin combination in mesothelioma.Expert Rev Anticancer Ther. 2005 Apr;5(2):231-7. doi: 10.1586/14737140.5.2.231. Expert Rev Anticancer Ther. 2005. PMID: 15877521 Review.
Cited by
-
Drug-repositioning screening identified fludarabine and risedronic acid as potential therapeutic compounds for malignant pleural mesothelioma.Invest New Drugs. 2021 Jun;39(3):644-657. doi: 10.1007/s10637-020-01040-y. Epub 2020 Dec 9. Invest New Drugs. 2021. PMID: 33300108 Free PMC article.
-
Genetic screens reveal new targetable vulnerabilities in BAP1-deficient mesothelioma.Cell Rep Med. 2023 Feb 21;4(2):100915. doi: 10.1016/j.xcrm.2022.100915. Epub 2023 Jan 18. Cell Rep Med. 2023. PMID: 36657447 Free PMC article.
-
Cellular Fitness Phenotypes of Cancer Target Genes from Oncobiology to Cancer Therapeutics.Cells. 2021 Feb 18;10(2):433. doi: 10.3390/cells10020433. Cells. 2021. PMID: 33670680 Free PMC article.
References
-
- Zalcman G, Mazieres J, Margery J, Greillier L, Audigier-Valette C, Moro-Sibilot D, et al. Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. Lancet. 2016;387:1405–1414. doi: 10.1016/S0140-6736(15)01238-6. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous