Pumilacidins from the Octocoral-Associated Bacillus sp. DT001 Display Anti-Proliferative Effects in Plasmodium falciparum
- PMID: 30158478
- PMCID: PMC6225264
- DOI: 10.3390/molecules23092179
Pumilacidins from the Octocoral-Associated Bacillus sp. DT001 Display Anti-Proliferative Effects in Plasmodium falciparum
Abstract
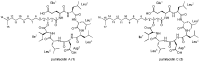
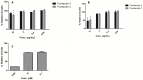
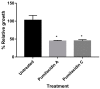
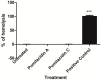
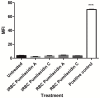
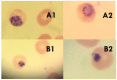
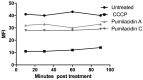
Chemical examination of the octocoral-associated Bacillus species (sp.) DT001 led to the isolation of pumilacidins A (1) and C (2). We investigated the effect of these compounds on the viability of Plasmodium falciparum and the mechanism of pumilacidin-induced death. The use of inhibitors of protein kinase C (PKC) and phosphoinositide 3-kinase (PI3K) was able to prevent the effects of pumilacidins A and C. The results indicated also that pumilacidins inhibit parasite growth via mitochondrial dysfunction and decreased cytosolic Ca2+.
Keywords: Bacillus; P. falciparum; apoptosis; coral-associated bacteria; pumilacidins; surfactins.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
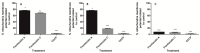


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

