Injective mesenchymal stem cell-based treatments for knee osteoarthritis: from mechanisms of action to current clinical evidences
- PMID: 30159741
- PMCID: PMC6541568
- DOI: 10.1007/s00167-018-5118-9
Injective mesenchymal stem cell-based treatments for knee osteoarthritis: from mechanisms of action to current clinical evidences
Abstract
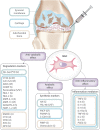
Purpose: Osteoarthritis (OA) represents a relevant social and economic burden worldwide. "Mesenchymal stem cells" or, as recently proposed, "medicinal signaling cells" (MSCs) have been recently introduced as injective treatments for OA with the aim of restoring joint homeostasis. The aim of this review is to provide the reader with the tools necessary to interpret the currently available clinical data, focusing on the MSC mechanisms of action which might help to clarify what we should expect from this treatment.
Methods: Clinical studies reporting MSC injections for the treatment of knee OA, either freshly isolated or culture-expanded cells, have been included and commented in relation to the supposed therapeutic effect that MSCs might exert giving their supposed mode of actions.
Results: The majority of the studies reports significant improvements in terms of pain and knee function compared to baseline values, up to 24 months of follow-up. Although these data support the expected therapeutic effect of this therapy giving the features of these cells, only 14% of the studies present a control group and more than one-third of them report the results on less than ten patients.
Conclusions: Despite the constant presence of positive and satisfactory results in the studies analyzed, the complexity of MSC metabolism and related therapeutic effects as well as the weakness of most of the studies do not allow withdrawing definitive conclusions about the superiority of one tissue source over another, as well as about the best cell dose and the long-term durability of the effects of these procedures. Given the high potential value of these therapies in the treatment of OA, further studies accurately designed, carefully defining the type of patients to be included and pursuing minimal standard requirements in terms of follow-up, number of patients, and types of measurements should be conducted to finally assess the efficacy of MSC-based injective treatments.
Keywords: Adipose tissue; Articular cartilage; Bone marrow concentrate; Inflammation; Intra-articular injection; Mesenchymal stem cells; Osteoarthritis; Stromal vascular fraction.
Conflict of interest statement
Laura de Girolamo is scientific consultant for Lipogems International S.p.A. The other authors declare that they do not have conflict of interests.
Figures

References
-
- Hiligsmann M, Cooper C, Arden N, Boers M, Branco JC, Luisa Brandi M, et al. Health economics in the field of osteoarthritis: an expert’s consensus paper from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) Semin Arthritis Rheum. 2013;43:303–313. doi: 10.1016/j.semarthrit.2013.07.003. - DOI - PubMed
-
- Paxton EW, Namba RS, Maletis GB, Khatod M, Yue EJ, Davies M, et al. A prospective study of 80,000 total joint and 5000 anterior cruciate ligament reconstruction procedures in a community-based registry in the United States. J Bone Joint Surg Am. 2010;92(Suppl 2):117–132. doi: 10.2106/JBJS.J.00807. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

