Identification of a GNE homozygous mutation in a Han-Chinese family with GNE myopathy
- PMID: 30160005
- PMCID: PMC6201217
- DOI: 10.1111/jcmm.13827
Identification of a GNE homozygous mutation in a Han-Chinese family with GNE myopathy
Abstract
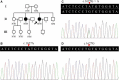
GNE myopathy is a rare, recessively inherited, early adult-onset myopathy, characterized by distal and proximal muscle degeneration which often spares the quadriceps. It is caused by mutations in the UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase gene (GNE). This study aimed to identify the disease-causing mutation in a three-generation Han-Chinese family with members who have been diagnosed with myopathy. A homozygous missense mutation, c.1627G>A (p.V543M) in the GNE gene co-segregates with the myopathy present in this family. A GNE myopathy diagnosis is evidenced by characteristic clinical manifestations, rimmed vacuoles in muscle biopsies and the presence of biallelic GNE mutations. This finding broadens the GNE gene mutation spectrum and extends the GNE myopathy phenotype spectrum.
Keywords: GNE myopathy; homozygous; missense mutation; the GNE gene.
© 2018 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures


Similar articles
-
Muscle biopsy and UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase gene mutation analysis in two Chinese patients with distal myopathy with rimmed vacuoles.Neuroreport. 2015 Jul 8;26(10):598-601. doi: 10.1097/WNR.0000000000000396. Neuroreport. 2015. PMID: 26053703
-
Analysis of the pathogenicity of novel GNE mutations and clinical, pathological, and genetic characteristics of GNE myopathy in Chinese population.Orphanet J Rare Dis. 2025 Apr 5;20(1):161. doi: 10.1186/s13023-025-03696-2. Orphanet J Rare Dis. 2025. PMID: 40188109 Free PMC article.
-
A Gne knockout mouse expressing human GNE D176V mutation develops features similar to distal myopathy with rimmed vacuoles or hereditary inclusion body myopathy.Hum Mol Genet. 2007 Nov 15;16(22):2669-82. doi: 10.1093/hmg/ddm220. Epub 2007 Aug 18. Hum Mol Genet. 2007. PMID: 17704511
-
Mutation update for GNE gene variants associated with GNE myopathy.Hum Mutat. 2014 Aug;35(8):915-26. doi: 10.1002/humu.22583. Hum Mutat. 2014. PMID: 24796702 Free PMC article. Review.
-
[GNE myopathy].Med Sci (Paris). 2015 Nov;31 Spec No 3:20-7. doi: 10.1051/medsci/201531s306. Epub 2015 Nov 6. Med Sci (Paris). 2015. PMID: 26546927 Review. French.
Cited by
-
Clinical, genetic, and pathological characterization of GNE myopathy in China.Neurol Sci. 2022 Jul;43(7):4483-4491. doi: 10.1007/s10072-022-05938-8. Epub 2022 Feb 9. Neurol Sci. 2022. PMID: 35138478
-
The role of amyloid β in the pathological mechanism of GNE myopathy.Neurol Sci. 2022 Nov;43(11):6309-6321. doi: 10.1007/s10072-022-06301-7. Epub 2022 Jul 29. Neurol Sci. 2022. PMID: 35904705 Free PMC article. Review.
-
Novel compound heterozygous mutations in a GNE myopathy with congenital thrombocytopenia: A case report and literature review.Clin Case Rep. 2022 Apr 4;10(4):e05659. doi: 10.1002/ccr3.5659. eCollection 2022 Apr. Clin Case Rep. 2022. PMID: 35414913 Free PMC article.
-
High expression of eukaryotic initiation factor 3M predicts poor prognosis in colon adenocarcinoma patients.Oncol Lett. 2020 Jan;19(1):876-884. doi: 10.3892/ol.2019.11164. Epub 2019 Nov 29. Oncol Lett. 2020. PMID: 31897202 Free PMC article.
-
Expanding the clinicopathological-genetic spectrum of GNE myopathy by a Chinese neuromuscular centre.J Cell Mol Med. 2021 Nov;25(22):10494-10503. doi: 10.1111/jcmm.16978. Epub 2021 Oct 22. J Cell Mol Med. 2021. PMID: 34676965 Free PMC article.
References
-
- Zhao J, Wang Z, Hong D, et al. Mutational spectrum and clinical features in 35 unrelated mainland Chinese patients with GNE myopathy. J Neurol Sci. 2015;354:21‐26. - PubMed
-
- No D, Valles‐Ayoub Y, Carbajo R, et al. Novel GNE mutations in autosomal recessive hereditary inclusion body myopathy patients. Genet Test Mol Biomarkers. 2013;17:376‐382. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

