Adverse drug reaction reporting among physicians working in private and government hospitals in Kuwait
- PMID: 30166908
- PMCID: PMC6111136
- DOI: 10.1016/j.jsps.2017.09.002
Adverse drug reaction reporting among physicians working in private and government hospitals in Kuwait
Abstract
Introduction: To improve patient safety and care, the identification and reporting of adverse drug reactions (ADRs) should be systematic and mandatory for all healthcare professionals (HCPs). Physicians remain the main HCPs with direct patient care whose role in ADRs reporting should not be ignored.
Objective: To document the awareness and attitude of physicians working in private and government hospitals in Kuwait with regard to pharmacovigilance (PV) and ADR reporting and to identify their practices of reporting ADRs.
Material and methods: A cross-sectional study was conducted using a paper-based 25-item questionnaire. The Statistical Package for Social Science (SPSS) was used for data analysis.
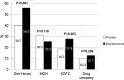
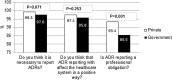
Results: A total of 1017 questionnaires were distributed to the eligible physicians in the government and private hospitals, giving a response rate of 84.2% and 83.0%, respectively (an overall response rate of 83.8%). Private physicians exhibited a better knowledge profile with regards to the purpose of PV (75.2% vs 64.8%; p = 0.002) and the correct ADR definition (75.8% vs 65.3%; p = 0.001). The majority of physicians showed good attitude towards reporting ADRs, nevertheless, private physicians had a significantly stronger belief that reporting ADRs is a professional obligation (93.4% vs 85.5%; p = 0.001). Three quarters of the study population (74.6%) had identified an ADR during their daily practice, however, only a small proportion (34.2%) confirms having ever reported ADRs. Regardless, significantly more private physicians had done so (42.4% vs 29.6%; p < 0.001). ADR reporting was significantly higher in physicians who knew the correct ADRs to be reported (adjusted OR = 1.86, p = 0.036), and those who were aware of any center or ADR reporting system in Kuwait (adjusted OR = 2.88, p = 0.020).
Conclusions: A national PV center empowered by clear legislation on "how" and "what" to report should improve physicians' reporting practices and hence is required in the country. This should be combined with constant training and education in this regard.
Keywords: Adverse drug reactions; Government; Hospitals; Pharmacovigilance; Physicians; Private.
Figures


Similar articles
-
Reporting of Adverse Drug Reactions in Primary Care Settings in Kuwait: A Comparative Study of Physicians and Pharmacists.Med Princ Pract. 2018;27(1):30-38. doi: 10.1159/000487236. Epub 2018 Jan 29. Med Princ Pract. 2018. PMID: 29402876 Free PMC article.
-
Knowledge, attitude and practices of pharmacovigilance and adverse drug reaction reporting among pharmacists working in secondary and tertiary governmental hospitals in Kuwait.Saudi Pharm J. 2017 Sep;25(6):830-837. doi: 10.1016/j.jsps.2016.12.004. Epub 2016 Dec 26. Saudi Pharm J. 2017. PMID: 28951666 Free PMC article.
-
Exploring healthcare professionals' knowledge, attitude, and practices towards pharmacovigilance: a cross-sectional survey.J Pharm Policy Pract. 2021 Jan 4;14(1):5. doi: 10.1186/s40545-020-00287-3. J Pharm Policy Pract. 2021. PMID: 33397478 Free PMC article.
-
Knowledge, attitude, practice and barriers towards pharmacovigilance and adverse drug reactions reporting among healthcare professionals in Turkey: a systematic review.Curr Med Res Opin. 2022 Jan;38(1):145-154. doi: 10.1080/03007995.2021.1997287. Epub 2021 Nov 3. Curr Med Res Opin. 2022. PMID: 34694167
-
Evaluation of patient reporting of adverse drug reactions to the UK 'Yellow Card Scheme': literature review, descriptive and qualitative analyses, and questionnaire surveys.Health Technol Assess. 2011 May;15(20):1-234, iii-iv. doi: 10.3310/hta15200. Health Technol Assess. 2011. PMID: 21545758 Review.
Cited by
-
A qualitative study of the reasons for low patient safety incident reporting among Indonesian nurses.Rev Bras Enferm. 2023 Oct 9;76(4):e20220583. doi: 10.1590/0034-7167-2022-0583. eCollection 2023. Rev Bras Enferm. 2023. PMID: 37820144 Free PMC article.
-
A Survey of Pharmacists and Other Healthcare Professionals in Vietnam: Factors Influencing Knowledge and Attitudes Toward Reporting Adverse Drug Reactions.Hosp Pharm. 2024 Feb;59(1):56-69. doi: 10.1177/00185787231186506. Epub 2023 Aug 22. Hosp Pharm. 2024. PMID: 38223867 Free PMC article.
-
Defining the awareness and attitude of the clinicians through pharmacovigilance in Turkey.World J Clin Cases. 2023 Jul 16;11(20):4865-4873. doi: 10.12998/wjcc.v11.i20.4865. World J Clin Cases. 2023. PMID: 37583988 Free PMC article.
-
Knowledge and Attitude of Health-Care Professionals Toward Adverse Drug Reactions Reporting at King Saud Medical City.J Pharm Bioallied Sci. 2018 Jan-Mar;10(1):29-34. doi: 10.4103/jpbs.JPBS_234_17. J Pharm Bioallied Sci. 2018. PMID: 29657505 Free PMC article.
-
Reporting of Adverse Drug Reactions in Primary Care Settings in Kuwait: A Comparative Study of Physicians and Pharmacists.Med Princ Pract. 2018;27(1):30-38. doi: 10.1159/000487236. Epub 2018 Jan 29. Med Princ Pract. 2018. PMID: 29402876 Free PMC article.
References
-
- Aithal S., Hooli T., Varun H. Knowledge and attitude about adverse drug reaction reporting among doctors at a tertiary care hospital. Int. J. Pharm. Biol. Sci. 2014;5:108–113.
-
- Al-Jarallah K., Moussa M., Al-Khanfar K.F. The physician workforce in Kuwait to the year 2020. Int. J. Health Plann. Manage. 2010;25:49–62. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

