Sulfanilamide in solution and liposome vesicles; in vitro release and UV-stability studies
- PMID: 30166909
- PMCID: PMC6111141
- DOI: 10.1016/j.jsps.2017.09.003
Sulfanilamide in solution and liposome vesicles; in vitro release and UV-stability studies
Abstract
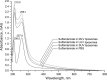
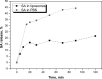
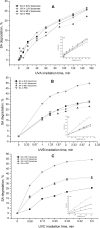
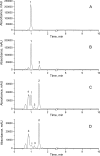
The main goal of this study was to develop a liposome formulation with sulfanilamide and to investigate the liposomes impact on its release and stability to the UV-A/UV-B and UV-C irradiation. Liposome dispersions with incorporated sulfanilamide were prepared by thin-film hydration method and liposomes role to the sulfanilamide release was investigated by using a dialysis method. Comparatively, sulfanilamide in phosphate buffer solution was subject to release study as well to the UV irradiation providing for the possibilities of kinetics analysis. In vitro drug release study demonstrated that 20% of sulfanilamide was released from liposomes within 1 h that is approximately twice as slower as in the case of dissolved sulfanilamide in phosphate buffer solution. The kinetic release process can be described by Korsmeyer-Peppas model and according to the value of diffusion release exponent it can be concluded that drug release mechanism is based on the phenomenon of diffusion. The sulfanilamide degradation in phosphate buffer solution and liposomes is related to the formation of UV-induced degradation products that are identified by UHPLC/MS analysis as: sulfanilic acid, aniline and benzidine. The UV-induced sulfanilamide degradation in the phosphate buffer solution and liposome vesicles fits the first- order kinetic model. The degradation rate constants are dependent on the involved UV photons energy input as well as sulfanilamide microenvironment. Liposome microenvironment provides better irradiation sulfanilamide stability. The obtained results suggest that liposomes might be promising carriers for delayed sulfanilamide delivery and may serve as a basis for further research.
Keywords: Degradation; Irradiation; Liposomes; Release; Stability; Sulfanilamide.
Figures
Similar articles
-
Intraliposomal chemical activation patterns of liposomal cis-bis-neodecanoato-trans-R,R-1,2-diaminocyclohexane platinum (II) (L-NDDP)-a potential antitumour agent.J Microencapsul. 2000 May-Jun;17(3):307-22. doi: 10.1080/026520400288283. J Microencapsul. 2000. PMID: 10819419
-
Investigation of liposomes as carriers of sodium ascorbyl phosphate for cutaneous photoprotection.Int J Pharm. 2005 Mar 3;291(1-2):21-9. doi: 10.1016/j.ijpharm.2004.07.039. Epub 2004 Dec 29. Int J Pharm. 2005. PMID: 15707728
-
A comparison between conventional liposome and drug-cyclodextrin complex in liposome system.Int J Pharm. 2016 Nov 20;513(1-2):387-392. doi: 10.1016/j.ijpharm.2016.09.043. Epub 2016 Sep 15. Int J Pharm. 2016. PMID: 27640244
-
Pharmacology of drugs formulated with DepoFoam: a sustained release drug delivery system for parenteral administration using multivesicular liposome technology.Clin Pharmacokinet. 2006;45(12):1153-76. doi: 10.2165/00003088-200645120-00002. Clin Pharmacokinet. 2006. PMID: 17112293 Review.
-
Liposomes for Pulmonary Drug Delivery: The Role of Formulation and Inhalation Device Design.Curr Pharm Des. 2017;23(3):362-372. doi: 10.2174/1381612823666161116114732. Curr Pharm Des. 2017. PMID: 27848886 Review.
Cited by
-
Stealth Liposomes (PEGylated) Containing an Anticancer Drug Camptothecin: In Vitro Characterization and In Vivo Pharmacokinetic and Tissue Distribution Study.Molecules. 2022 Feb 6;27(3):1086. doi: 10.3390/molecules27031086. Molecules. 2022. PMID: 35164350 Free PMC article.
-
Rosehip Extract-Loaded Liposomes for Potential Skin Application: Physicochemical Properties of Non- and UV-Irradiated Liposomes.Plants (Basel). 2023 Aug 25;12(17):3063. doi: 10.3390/plants12173063. Plants (Basel). 2023. PMID: 37687310 Free PMC article.
-
Development of a novel intramuscular liposomal injection for advanced meloxicam delivery: Preparation, characterization, in vivo pharmacokinetics, pharmacodynamics, and pain assessment in an orthopedic pain model.Int J Pharm X. 2024 Sep 14;8:100284. doi: 10.1016/j.ijpx.2024.100284. eCollection 2024 Dec. Int J Pharm X. 2024. PMID: 39323733 Free PMC article.
-
Synergism of cisplatin-oleanolic acid co-loaded calcium carbonate nanoparticles on hepatocellular carcinoma cells for enhanced apoptosis and reduced hepatotoxicity.Int J Nanomedicine. 2019 May 28;14:3753-3771. doi: 10.2147/IJN.S196651. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31239661 Free PMC article.
-
Novel Findings about Double-Loaded Curcumin-in-HPβcyclodextrin-in Liposomes: Effects on the Lipid Bilayer and Drug Release.Pharmaceutics. 2018 Dec 3;10(4):256. doi: 10.3390/pharmaceutics10040256. Pharmaceutics. 2018. PMID: 30513858 Free PMC article.
References
-
- Albini A., Fasani E. In: Photostability of Drugs and Drug Formulations. Tønnesen H.H., editor. CRC Press; Boca Raton: 2004. Rationalizing the photochemistry of drugs; pp. 67–111.
-
- Baran W., Sochacka J., Wardas W. Toxicity and biodegradability of sulfonamides and products of their photocatalytic degradation in aqueous solutions. Chemosphere. 2006;65(8):1295–1299. - PubMed
-
- Boreen A.L., Arnold W.A., McNeill K. Photochemical fate of sulfa drugs in the aquatic environment: sulfa drugs containing five-membered heterocyclic groups. Environ. Sci. Technol. 2004;38(14):3933–3940. - PubMed
-
- Brown C.K., Friedel H.D., Barker A.R., Buhse L.F., Keitel S., Cecil T.L., Kraemer J., Morris M., Reppas C., Stickelmeyer M., Shah V., Yomota C. FIP/AAPS joint workshop report: dissolution/in vitro release testing of novel/special dosage forms. Indian. J. Pharm. Sci. 2011;73(3):338–353. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

