Genetic and Molecular Basis of Feather Diversity in Birds
- PMID: 30169786
- PMCID: PMC6171735
- DOI: 10.1093/gbe/evy180
Genetic and Molecular Basis of Feather Diversity in Birds
Abstract
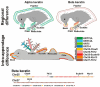
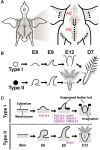
Feather diversity is striking in many aspects. Although the development of feather has been studied for decades, genetic and genomic studies of feather diversity have begun only recently. Many questions remain to be answered by multidisciplinary approaches. In this review, we discuss three levels of feather diversity: Feather morphotypes, intraspecific variations, and interspecific variations. We summarize recent studies of feather evolution in terms of genetics, genomics, and developmental biology and provide perspectives for future research. Specifically, this review includes the following topics: 1) Diversity of feather morphotype; 2) feather diversity among different breeds of domesticated birds, including variations in pigmentation pattern, in feather length or regional identity, in feather orientation, in feather distribution, and in feather structure; and 3) diversity of feathers among avian species, including plumage color and morph differences between species and the regulatory differences in downy feather development between altricial and precocial birds. Finally, we discussed future research directions.
Figures



Similar articles
-
Variations in the morphology, distribution, and arrangement of feathers in domesticated birds.J Exp Zool B Mol Dev Evol. 2003 Aug 15;298(1):91-108. doi: 10.1002/jez.b.28. J Exp Zool B Mol Dev Evol. 2003. PMID: 12949771 Review.
-
Feather Evolution from Precocial to Altricial Birds.Zool Stud. 2019 Sep 16;58:e24. doi: 10.6620/ZS.2019.58-24. eCollection 2019. Zool Stud. 2019. PMID: 31966325 Free PMC article.
-
Complex plumages spur rapid color diversification in kingfishers (Aves: Alcedinidae).Elife. 2023 Apr 21;12:e83426. doi: 10.7554/eLife.83426. Elife. 2023. PMID: 37083474 Free PMC article.
-
Reaction-diffusion models of within-feather pigmentation patterning.Proc Biol Sci. 2002 Apr 22;269(1493):781-92. doi: 10.1098/rspb.2001.1896. Proc Biol Sci. 2002. PMID: 11958709 Free PMC article.
-
Diversity, physiology, and evolution of avian plumage carotenoids and the role of carotenoid-protein interactions in plumage color appearance.Arch Biochem Biophys. 2015 Apr 15;572:201-212. doi: 10.1016/j.abb.2015.01.016. Epub 2015 Jan 28. Arch Biochem Biophys. 2015. PMID: 25637658 Review.
Cited by
-
Genome-Wide Association Study Identifies Candidate Genes for Stripe Pattern Feather Color of Rhode Island Red Chicks.Genes (Basel). 2022 Aug 24;13(9):1511. doi: 10.3390/genes13091511. Genes (Basel). 2022. PMID: 36140679 Free PMC article.
-
In Ovo Injection of CHIR-99021 Promotes Feather Follicle Development via Modulating the Wnt Signaling Pathway and Transcriptome in Goose Embryos (Anser cygnoides).Front Physiol. 2022 May 20;13:858274. doi: 10.3389/fphys.2022.858274. eCollection 2022. Front Physiol. 2022. PMID: 35669574 Free PMC article.
-
Identification of Polymorphisms in the HERC2-OCA2 Gene Locus and their Association with Feather Color in Quail.J Poult Sci. 2023 May 25;60(2):2023013. doi: 10.2141/jpsa.2023013. eCollection 2023. J Poult Sci. 2023. PMID: 37234755 Free PMC article.
-
The First Crested Duck Genome Reveals Clues to Genetic Compensation and Crest Cushion Formation.Genomics Proteomics Bioinformatics. 2023 Jun;21(3):483-500. doi: 10.1016/j.gpb.2023.08.002. Epub 2023 Aug 29. Genomics Proteomics Bioinformatics. 2023. PMID: 37652165 Free PMC article.
-
Transcriptional Characteristics Showed That miR-144-y/FOXO3 Participates in Embryonic Skin and Feather Follicle Development in Zhedong White Goose.Animals (Basel). 2022 Aug 17;12(16):2099. doi: 10.3390/ani12162099. Animals (Basel). 2022. PMID: 36009690 Free PMC article.
References
-
- Abbott UK, Asmundson VS.. 1957. Scaleless, an inherited ectodermal defect in the domestic fowl. J Hered. 48(2):63–70.
-
- Alibardi L. 2016. Review: cornification, morphogenesis and evolution of feathers. Protoplasma 254(3):1259–1281. - PubMed
-
- Auber L. 1941. The colours of feathers and their structural causes in varieties of the Budgerigar, Melopsittacus undulatus (Shaw). Edinburgh (United Kingdom: ): University of Edinburgh.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

