Functional and Mechanistic Neurotoxicity Profiling Using Human iPSC-Derived Neural 3D Cultures
- PMID: 30169818
- PMCID: PMC6317428
- DOI: 10.1093/toxsci/kfy218
Functional and Mechanistic Neurotoxicity Profiling Using Human iPSC-Derived Neural 3D Cultures
Abstract
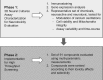
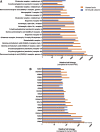
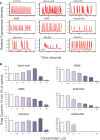
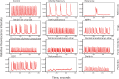
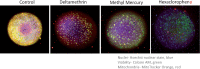
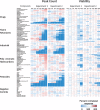
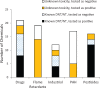
Neurological disorders affect millions of people worldwide and appear to be on the rise. Whereas the reason for this increase remains unknown, environmental factors are a suspected contributor. Hence, there is an urgent need to develop more complex, biologically relevant, and predictive in vitro assays to screen larger sets of compounds with the potential for neurotoxicity. Here, we employed a human induced pluripotent stem cell (iPSC)-based 3D neural platform composed of mature cortical neurons and astrocytes as a model for this purpose. The iPSC-derived human 3D cortical neuron/astrocyte co-cultures (3D neural cultures) present spontaneous synchronized, readily detectable calcium oscillations. This advanced neural platform was optimized for high-throughput screening in 384-well plates and displays highly consistent, functional performance across different wells and plates. Characterization of oscillation profiles in 3D neural cultures was performed through multi-parametric analysis that included the calcium oscillation rate and peak width, amplitude, and waveform irregularities. Cellular and mitochondrial toxicity were assessed by high-content imaging. For assay characterization, we used a set of neuromodulators with known mechanisms of action. We then explored the neurotoxic profile of a library of 87 compounds that included pharmaceutical drugs, pesticides, flame retardants, and other chemicals. Our results demonstrated that 57% of the tested compounds exhibited effects in the assay. The compounds were then ranked according to their effective concentrations based on in vitro activity. Our results show that a human iPSC-derived 3D neural culture assay platform is a promising biologically relevant tool to assess the neurotoxic potential of drugs and environmental toxicants.
Figures



References
-
- Abou-Donia M. B. (2003). Organophosphorus ester-induced chronic neurotoxicity. Arche. Environ. Health 58, 484–497. - PubMed
-
- Arvanov V. L., Liang X., Schwartz J., Grossman S., Wang R. Y. (1997). Clozapine and haloperidol modulate N-methyl-d-aspartate- and non-N-methyl-d-aspartate receptor-mediated neurotransmission in rat prefrontal cortical neurons in vitro. J. Pharmacol. Exp. Ther. 283, 226–234. - PubMed
-
- Beattie E. C., Carroll R. C., Yu X., Morishita W., Yasuda H., von Zastrow M., Malenka R. C. (2000). Regulation of AMPA receptor endocytosis by a signaling mechanism shared with LTD. Nat. Neurosci. 3, 1291–1300. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

