Anticancer Activity of Metformin, an Antidiabetic Drug, Against Ovarian Cancer Cells Involves Inhibition of Cysteine-Rich 61 (Cyr61)/Akt/Mammalian Target of Rapamycin (mTOR) Signaling Pathway
- PMID: 30171812
- PMCID: PMC6130173
- DOI: 10.12659/MSM.909745
Anticancer Activity of Metformin, an Antidiabetic Drug, Against Ovarian Cancer Cells Involves Inhibition of Cysteine-Rich 61 (Cyr61)/Akt/Mammalian Target of Rapamycin (mTOR) Signaling Pathway
Retraction in
-
Retracted: Anticancer Activity of Metformin, an Antidiabetic Drug, Against Ovarian Cancer Cells Involves Inhibition of Cysteine-Rich 61 (Cyr61)/Akt/Mammalian Target of Rapamycin (mTOR) Signaling Pathway.Med Sci Monit. 2021 Mar 25;27:e932331. doi: 10.12659/MSM.932331. Med Sci Monit. 2021. PMID: 33762564 Free PMC article.
Abstract
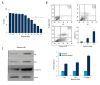
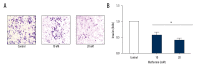
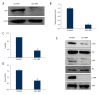
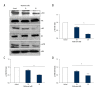
BACKGROUND Ovarian cancer is considered one of the lethal cancers responsible for high mortality and morbidity across the world. The prognosis and the survival rate of ovarian cancer is far from decent. Cysteine-rich 61 (Cyr61) also known as CCN1, is a member of CCN-family of growth factors, reported to be significantly overexpressed in several malignancies which include, but are not limited to, ovarian cancer. Recent studies have revealed that women with type 2 diabetes mellitus have an elevated risk of ovarian cancer. Hence, administration of an antidiabetic drug with anticancer effects such as metformin may act as an effective therapeutic regime against ovarian cancer. MATERIAL AND METHODS Cell viability and apoptosis were examined by MTT and Annexin V/PI double staining respectively. Cell migration was determined by Boyden Chamber assay. Transient knockdown of Cyr61 in ovarian cancer cells was achieved by transecting the cells with siRNA for Cyr61using Lipofectamine 2000. RESULTS Our results indicated that treatment of ovarian cancer cells with metformin caused significant downregulation of Cyr61 protein expression levels ultimately favoring apoptosis. Transient knockdown of Cyr61 resulted in the inhibition of cell proliferation and migration. This was also associated with the concomitant downregulation of pAkt and pmTOR confirming the role of Cyr61 as an upstream modulator of Akt signaling. Conversely the extracellular supplementation of recombinant Cyr61 attenuates the cytotoxic properties of metformin in ovarian cancer cells. CONCLUSIONS Taken together, we concluded that metformin exhibits anticancer effects and Cyr61 acts as a direct target for metformin in ovarian cancer cells.
Figures

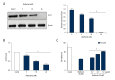
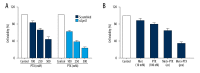
References
-
- Jayson GC, Kohn EC, Kitchener HC, Ledermann JA. Ovarian cancer. Lancet. 2014;384(9951):1376–88. - PubMed
-
- Nam MS, Jung DB, Seo KH, et al. Apoptotic effect of Sanggenol L via caspase activation and inhibition of NF-kappaB signaling in ovarian cancer cells. Phytother Res. 2016;30(1):90–96. - PubMed
-
- Ivanov S, Ivanov S, Khadzhiolov N. [Ovarian tumours – accuracy of frozen section diagnosis]. Akush Ginekol (Sofiia) 2005;44(1):11–13. [in Bulgarian] - PubMed
-
- Saika K, Sobue T. [Cancer statistics in the world]. Gan To Kagaku Ryoho. 2013;40(13):2475–80. [in Japanese] - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

