Downstaging and Survival Outcomes Associated With Neoadjuvant Chemotherapy Regimens Among Patients Treated With Cystectomy for Muscle-Invasive Bladder Cancer
- PMID: 30178038
- PMCID: PMC6248089
- DOI: 10.1001/jamaoncol.2018.3542
Downstaging and Survival Outcomes Associated With Neoadjuvant Chemotherapy Regimens Among Patients Treated With Cystectomy for Muscle-Invasive Bladder Cancer
Abstract
Importance: Neoadjuvant chemotherapy (NAC) followed by radical cystectomy improves survival compared with cystectomy alone for patients with bladder cancer. Although gemcitabine with cisplatin has become a standard NAC regimen, a dose-dense combination of methotrexate, vinblastine, doxorubicin, and cisplatin (ddMVAC) is being adopted at some institutions.
Objective: To assess the association of neoadjuvant ddMVAC vs standard regimens with downstaging and overall survival among patients treated with radical cystectomy for bladder cancer.
Design, setting, and participants: Cross-sectional analysis of data extracted from the medical records of a consecutive sample, after exclusions, of 1113 patients with bladder cancer of whom 824 had disease stage T2 or greater, who were treated with cystectomy at the Moffitt Cancer Center in Tampa, Florida, a tertiary care cancer center, between January 1, 2007, and May 31, 2017. Data were collected between November 14, 2016, and July 21, 2017, and analyzed between August 21, 2017, and December 8, 2017. Patients were compared based on type of NAC. Those who did not receive NAC were included as controls.
Main outcomes and measures: Comparative rates and the association of any downstaging, complete response, and overall survival with ddMVAC and other NAC regimens and surgery alone. Outcomes were examined using Kaplan-Meier, adjusted logistic, Cox regression, and propensity-weighted models.
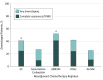
Results: Of the 1113 patients who underwent cystectomy for bladder cancer, 861 (77.4%) were male, the median (interquartile range) age was 67 (60-74) years, 1051 (94.4%) were white, 27 (2.4%) black, 37 (3.3%) Hispanic/Latino, and 35 (3.1%) other race/ethnicity. Of 824 patients with muscle-invasive bladder cancer, 332 (40%) received NAC. Downstaging rates were 52.2% for ddMVAC, 41.3% for gemcitabine-cisplatin, and 27.0% for gemcitabine with carboplatin, and complete response (pT0N0) rates were 41.3% for ddMVAC, 24.5% for gemcitabine-cisplatin, and 9.4% for gemcitabine-carboplatin (2-sided P < .001). Adjusted analysis comparing ddMVAC with gemcitabine-cisplatin demonstrated a higher likelihood of downstaging (odds ratio [OR], 1.84; 95% CI, 1.10-3.09) and complete response (OR, 2.67; 95% CI, 1.50-4.77) with ddMVAC. Similar results were achieved with propensity score matching (OR, 1.52; 95% CI, 0.99-2.35). Patients who received ddMVAC had better overall survival than those treated with other chemotherapy regimens, although the observed survival benefit did not reach statistical significance in adjusted or propensity-matched models (hazard ratio, 0.44; 95% CI, 0.14-1.38; P = .16).
Conclusions and relevance: This study suggest that neoadjuvant ddMVAC followed by cystectomy is associated with a higher complete response (ypT0N0) rate than standard NAC. These data highlight and suggest the need to further investigate ddMVAC vs standard NAC in a prospective, randomized fashion.
Conflict of interest statement
Figures


Comment in
-
Reply letter to: Peyton CC, Tang D, Reich RR, et al. Downstaging and survival outcomes associated with neoadjuvant chemotherapy regimens among patients treated with cystectomy for muscle-invasive bladder cancer. JAMA Oncol. 2018 Nov 1;4(11):1535-1542.World J Urol. 2020 May;38(5):1341-1342. doi: 10.1007/s00345-019-02751-5. Epub 2019 Apr 3. World J Urol. 2020. PMID: 30944970 No abstract available.
References
-
- Advanced Bladder Cancer (ABC) Meta-analysis Collaboration Neoadjuvant chemotherapy in invasive bladder cancer: update of a systematic review and meta-analysis of individual patient data advanced bladder cancer (ABC) meta-analysis collaboration. Eur Urol. 2005;48(2):202-205. doi:10.1016/j.eururo.2005.04.006 - DOI - PubMed
-
- Griffiths G, Hall R, Sylvester R, Raghavan D, Parmar MK; International Collaboration of Trialists; Medical Research Council Advanced Bladder Cancer Working Party (now the National Cancer Research Institute Bladder Cancer Clinical Studies Group); European Organisation for Research and Treatment of Cancer Genito-Urinary Tract Cancer Group; Australian Bladder Cancer Study Group; National Cancer Institute of Canada Clinical Trials Group; Finnbladder; Norwegian Bladder Cancer Study Group; Club Urologico Espanol de Tratamiento Oncologico Group . International phase III trial assessing neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: long-term results of the BA06 30894 trial. J Clin Oncol. 2011;29(16):2171-2177. doi:10.1200/JCO.2010.32.3139 - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

