Complement activation contributes to perioperative neurocognitive disorders in mice
- PMID: 30180861
- PMCID: PMC6123969
- DOI: 10.1186/s12974-018-1292-4
Complement activation contributes to perioperative neurocognitive disorders in mice
Abstract
Background: The complement system plays an important role in many neurological disorders. Complement modulation, including C3/C3a receptor signaling, shows promising therapeutic effects on cognition and neurodegeneration. Yet, the implications for this pathway in perioperative neurocognitive disorders (PND) are not well established. Here, we evaluated the possible role for C3/C3a receptor signaling after orthopedic surgery using an established mouse model of PND.
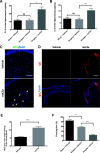
Methods: A stabilized tibial fracture surgery was performed in adult male C57BL/6 mice under general anesthesia and analgesia to induce PND-like behavior. Complement activation was assessed in the hippocampus and choroid plexus. Changes in hippocampal neuroinflammation, synapse numbers, choroidal blood-cerebrospinal fluid barrier (BCSFB) integrity, and hippocampal-dependent memory function were evaluated after surgery and treatment with a C3a receptor blocker.
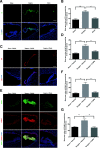
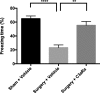
Results: C3 levels and C3a receptor expression were specifically increased in hippocampal astrocytes and microglia after surgery. Surgery-induced neuroinflammation and synapse loss in the hippocampus were attenuated by C3a receptor blockade. Choroidal BCSFB dysfunction occurred 1 day after surgery and was attenuated by C3a receptor blockade. Administration of exogenous C3a exacerbated cognitive decline after surgery, whereas C3a receptor blockade improved hippocampal-dependent memory function.
Conclusions: Orthopedic surgery activates complement signaling. C3a receptor blockade may be therapeutically beneficial to attenuate neuroinflammation and PND.
Keywords: Choroid plexus; Complement; Hippocampus; Neuroinflammation; Perioperative neurocognitive disorders.
Conflict of interest statement
Ethics approval and consent to participate
All procedures were approved by Institutional Animal Care and Use Committee at Capital Medical University (Beijing, China) and carried out under the rules of Medical Research Center of Beijing Chao-Yang Hospital (Beijing, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
IL-17A contributes to perioperative neurocognitive disorders through blood-brain barrier disruption in aged mice.J Neuroinflammation. 2018 Nov 30;15(1):332. doi: 10.1186/s12974-018-1374-3. J Neuroinflammation. 2018. PMID: 30501622 Free PMC article.
-
Interleukin-33 ameliorates perioperative neurocognitive disorders by modulating microglial state.Neuropharmacology. 2024 Aug 1;253:109982. doi: 10.1016/j.neuropharm.2024.109982. Epub 2024 May 1. Neuropharmacology. 2024. PMID: 38701943
-
A complement-microglial axis driving inhibitory synapse related protein loss might contribute to systemic inflammation-induced cognitive impairment.Int Immunopharmacol. 2020 Oct;87:106814. doi: 10.1016/j.intimp.2020.106814. Epub 2020 Jul 21. Int Immunopharmacol. 2020. PMID: 32707491
-
Postoperative analgesia with morphine promoting microglial activation and neuroinflammation induced by surgery aggravates perioperative neurocognitive dysfunction in aged mice.IBRO Neurosci Rep. 2024 Dec 17;18:39-49. doi: 10.1016/j.ibneur.2024.12.008. eCollection 2025 Jun. IBRO Neurosci Rep. 2024. PMID: 39816480 Free PMC article. Review.
-
Neuroinflammation in perioperative neurocognitive disorders: From bench to the bedside.CNS Neurosci Ther. 2022 Apr;28(4):484-496. doi: 10.1111/cns.13794. Epub 2022 Jan 6. CNS Neurosci Ther. 2022. PMID: 34990087 Free PMC article. Review.
Cited by
-
SIRT1 activation attenuates microglia-mediated synaptic engulfment in postoperative cognitive dysfunction.Front Aging Neurosci. 2022 Nov 10;14:943842. doi: 10.3389/fnagi.2022.943842. eCollection 2022. Front Aging Neurosci. 2022. PMID: 36437988 Free PMC article.
-
Role of gut microbiota in neuroinflammation: a focus on perioperative neurocognitive disorders.Front Cell Infect Microbiol. 2025 Jul 7;15:1582909. doi: 10.3389/fcimb.2025.1582909. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40692688 Free PMC article. Review.
-
Peripheral inflammation as a potential mechanism and preventive strategy for perioperative neurocognitive disorder under general anesthesia and surgery.Front Cell Neurosci. 2024 Jul 3;18:1365448. doi: 10.3389/fncel.2024.1365448. eCollection 2024. Front Cell Neurosci. 2024. PMID: 39022312 Free PMC article. Review.
-
Postoperative cognitive dysfunction in the aged: the collision of neuroinflammaging with perioperative neuroinflammation.Inflammopharmacology. 2019 Feb;27(1):27-37. doi: 10.1007/s10787-018-00559-0. Epub 2019 Jan 3. Inflammopharmacology. 2019. PMID: 30607668 Review.
-
The biological alterations of synapse/synapse formation in sepsis-associated encephalopathy.Front Synaptic Neurosci. 2022 Dec 2;14:1054605. doi: 10.3389/fnsyn.2022.1054605. eCollection 2022. Front Synaptic Neurosci. 2022. PMID: 36530954 Free PMC article. Review.
References
-
- Inouye SK, Marcantonio ER, Kosar CM, Tommet D, Schmitt EM, Travison TG, Saczynski JS, Ngo LH, Alsop DC, Jones RN. The short-term and long-term relationship between delirium and cognitive trajectory in older surgical patients. Alzheimers Dement. 2016;12:766–775. doi: 10.1016/j.jalz.2016.03.005. - DOI - PMC - PubMed
-
- Evered L, Silbert B, Knopman DS, Scott DA, ST DK, Rasmussen LS, Oh ES, Crosby G, Berger M, Eckenhoff RG, Group. TNCW. Recommendations for the nomenclature of cognitive change associated with anaesthesia and surgery. Br J Anaesth. 2018; In press.
-
- Skvarc DR, Berk M, Byrne LK, Dean OM, Dodd S, Lewis M, Marriott A, Moore EM, Morris G, Page RS, Gray L. Post-operative cognitive dysfunction: an exploration of the inflammatory hypothesis and novel therapies. Neurosci Biobehav Rev. 2018;84:116–33. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

