Notch signaling mediated by Delta-like ligands 1 and 4 controls the pathogenesis of chronic GVHD in mice
- PMID: 30181175
- PMCID: PMC6238189
- DOI: 10.1182/blood-2018-03-841155
Notch signaling mediated by Delta-like ligands 1 and 4 controls the pathogenesis of chronic GVHD in mice
Abstract
Chronic graft-versus-host disease (cGVHD) is a major complication of allogeneic hematopoietic cell transplantation (allo-HCT) and remains an area of unmet clinical need with few treatment options available. Notch blockade prevents acute GVHD in multiple mouse models, but the impact of Notch signaling on cGVHD remains unknown. Using genetic and antibody-mediated strategies of Notch inhibition, we investigated the role of Notch signaling in complementary mouse cGVHD models that mimic several aspects of human cGVHD in search of candidate therapeutics. In the B10.D2→BALB/c model of sclerodermatous cGVHD, Delta-like ligand 4 (Dll4)-driven Notch signaling was essential for disease development. Antibody-mediated Dll4 inhibition conferred maximum benefits when pursued early in a preventative fashion, with anti-Dll1 enhancing early protection. Notch-deficient alloantigen-specific T cells showed no early defects in proliferation or helper polarization in vivo but subsequently exhibited markedly decreased cytokine secretion and enhanced accumulation of FoxP3+ regulatory T cells. In the B6→B10.BR major histocompatibility complex-mismatched model with multi-organ system cGVHD and prominent bronchiolitis obliterans (BO), but not skin manifestations, absence of Notch signaling in T cells provided long-lasting disease protection that was replicated by systemic targeting of Dll1, Dll4, or both Notch ligands, even during established disease. Notch inhibition decreased target organ damage and germinal center formation. Moreover, decreased BO-cGVHD was observed upon inactivation of Notch1 and/or Notch2 in T cells. Systemic targeting of Notch2 alone was safe and conferred therapeutic benefits. Altogether, Notch ligands and receptors regulate key pathogenic steps in cGVHD and emerge as novel druggable targets to prevent or treat different forms of cGVHD.
© 2018 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: M.Y. and C.W.S. are employees of Genentech, Inc. The remaining authors declare no competing financial interests.
Figures

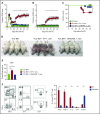
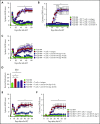



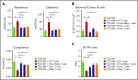

Comment in
-
Int"Dll"igent control of T-cell pathology in GVHD.Blood. 2018 Nov 15;132(20):2112-2114. doi: 10.1182/blood-2018-09-875120. Blood. 2018. PMID: 30442748 No abstract available.
Similar articles
-
Early Notch Signals Induce a Pathogenic Molecular Signature during Priming of Alloantigen-Specific Conventional CD4+ T Cells in Graft-versus-Host Disease.J Immunol. 2019 Jul 15;203(2):557-568. doi: 10.4049/jimmunol.1900192. Epub 2019 Jun 10. J Immunol. 2019. PMID: 31182480 Free PMC article.
-
Transient blockade of delta-like Notch ligands prevents allograft rejection mediated by cellular and humoral mechanisms in a mouse model of heart transplantation.J Immunol. 2015 Mar 15;194(6):2899-908. doi: 10.4049/jimmunol.1402034. Epub 2015 Feb 16. J Immunol. 2015. PMID: 25687759 Free PMC article.
-
T cells take directions from supporting cast in graft-versus-host disease.J Clin Invest. 2017 Apr 3;127(4):1215-1217. doi: 10.1172/JCI93552. Epub 2017 Mar 20. J Clin Invest. 2017. PMID: 28319046 Free PMC article.
-
Notch signaling in hematopoietic cell transplantation and T cell alloimmunity.Blood Rev. 2013 Nov;27(6):269-77. doi: 10.1016/j.blre.2013.08.001. Epub 2013 Aug 31. Blood Rev. 2013. PMID: 24050990 Free PMC article. Review.
-
[Recent advances in prevention and treatment of chronic graft-versus-host disease after allogeneic hematopoietic stem cell transplantation].Rinsho Ketsueki. 2024;65(5):401-411. doi: 10.11406/rinketsu.65.401. Rinsho Ketsueki. 2024. PMID: 38825520 Review. Japanese.
Cited by
-
Roles of Notch Signaling in the Tumor Microenvironment.Int J Mol Sci. 2022 Jun 2;23(11):6241. doi: 10.3390/ijms23116241. Int J Mol Sci. 2022. PMID: 35682918 Free PMC article. Review.
-
Early Notch Signals Induce a Pathogenic Molecular Signature during Priming of Alloantigen-Specific Conventional CD4+ T Cells in Graft-versus-Host Disease.J Immunol. 2019 Jul 15;203(2):557-568. doi: 10.4049/jimmunol.1900192. Epub 2019 Jun 10. J Immunol. 2019. PMID: 31182480 Free PMC article.
-
Chronic GvHD NIH Consensus Project Biology Task Force: evolving path to personalized treatment of chronic GvHD.Blood Adv. 2023 Sep 12;7(17):4886-4902. doi: 10.1182/bloodadvances.2022007611. Blood Adv. 2023. PMID: 36322878 Free PMC article.
-
National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2020 Etiology and Prevention Working Group Report.Transplant Cell Ther. 2021 Jun;27(6):452-466. doi: 10.1016/j.jtct.2021.02.035. Epub 2021 Mar 2. Transplant Cell Ther. 2021. PMID: 33877965 Free PMC article. Review.
-
Controversies and expectations for the prevention of GVHD: A biological and clinical perspective.Front Immunol. 2022 Nov 23;13:1057694. doi: 10.3389/fimmu.2022.1057694. eCollection 2022. Front Immunol. 2022. PMID: 36505500 Free PMC article. Review.
References
-
- Shlomchik WD. Graft-versus-host disease. Nat Rev Immunol. 2007;7(5):340-352. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

