FcαRI binding at the IgA1 CH2-CH3 interface induces long-range conformational changes that are transmitted to the hinge region
- PMID: 30181292
- PMCID: PMC6156678
- DOI: 10.1073/pnas.1807478115
FcαRI binding at the IgA1 CH2-CH3 interface induces long-range conformational changes that are transmitted to the hinge region
Abstract
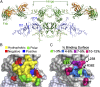
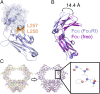
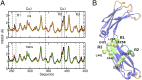
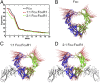
IgA effector functions include proinflammatory immune responses triggered upon clustering of the IgA-specific receptor, FcαRI, by IgA immune complexes. FcαRI binds to the IgA1-Fc domain (Fcα) at the CH2-CH3 junction and, except for CH2 L257 and L258, all side-chain contacts are contributed by the CH3 domain. In this study, we used experimental and computational approaches to elucidate energetic and conformational aspects of FcαRI binding to IgA. The energetic contribution of each IgA residue in the binding interface was assessed by alanine-scanning mutagenesis and equilibrium surface plasmon resonance (SPR). As expected, hydrophobic residues central to the binding site have strong energetic contributions to the FcαRI:Fcα interaction. Surprisingly, individual mutation of CH2 residues L257 and L258, found at the periphery of the FcαRI binding site, dramatically reduced binding affinity. Comparison of antibody:receptor complexes involving IgA or its precursor IgY revealed a conserved receptor binding site at the CH2-CH3 junction (or its equivalent). Given the importance of residues near the CH2-CH3 junction, we used coarse-grained Langevin dynamics simulations to understand the functional dynamics in Fcα. Our simulations indicate that FcαRI binding, either in an asymmetric (1:1) or symmetric (2:1) complex with Fcα, propagated long-range conformational changes across the Fc domains, potentially impacting the hinge and Fab regions. Subsequent SPR experiments confirmed that FcαRI binding to the Fcα CH2-CH3 junction altered the kinetics of HAA lectin binding at the IgA1 hinge. Receptor-induced long-distance conformational transitions have important implications for the interaction of aberrantly glycosylated IgA1 with anti-glycan autoantibodies in IgA nephropathy.
Keywords: IgA1 antibody; binding energetics; molecular-dynamics simulations; principal-component analysis; surface plasmon resonance.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Insights into IgA-mediated immune responses from the crystal structures of human FcalphaRI and its complex with IgA1-Fc.Nature. 2003 Jun 5;423(6940):614-20. doi: 10.1038/nature01685. Epub 2003 May 21. Nature. 2003. PMID: 12768205
-
Bivalent binding of IgA1 to FcalphaRI suggests a mechanism for cytokine activation of IgA phagocytosis.J Mol Biol. 2003 Mar 28;327(3):645-57. doi: 10.1016/s0022-2836(03)00149-9. J Mol Biol. 2003. PMID: 12634059
-
Analysis of IgA1 N-glycosylation and its contribution to FcalphaRI binding.Biochemistry. 2008 Oct 28;47(43):11285-99. doi: 10.1021/bi801185b. Epub 2008 Oct 1. Biochemistry. 2008. PMID: 18826328 Free PMC article.
-
IgA Fc receptors.Annu Rev Immunol. 2003;21:177-204. doi: 10.1146/annurev.immunol.21.120601.141011. Epub 2001 Dec 19. Annu Rev Immunol. 2003. PMID: 12524384 Review.
-
Role of IgA receptors in the pathogenesis of IgA nephropathy.J Nephrol. 2016 Feb;29(1):5-11. doi: 10.1007/s40620-015-0246-5. Epub 2015 Nov 14. J Nephrol. 2016. PMID: 26572664 Review.
Cited by
-
Engineering a pure and stable heterodimeric IgA for the development of multispecific therapeutics.MAbs. 2022 Jan-Dec;14(1):2141637. doi: 10.1080/19420862.2022.2141637. MAbs. 2022. PMID: 36343329 Free PMC article.
-
Effects of N-glycans on the structure of human IgA2.Front Mol Biosci. 2024 Apr 5;11:1390659. doi: 10.3389/fmolb.2024.1390659. eCollection 2024. Front Mol Biosci. 2024. PMID: 38645274 Free PMC article.
-
Mechanism and inhibition of Streptococcus pneumoniae IgA1 protease.Nat Commun. 2020 Nov 27;11(1):6063. doi: 10.1038/s41467-020-19887-3. Nat Commun. 2020. PMID: 33247098 Free PMC article.
-
Distinct Fcα receptor N-glycans modulate the binding affinity to immunoglobulin A (IgA) antibodies.J Biol Chem. 2019 Sep 20;294(38):13995-14008. doi: 10.1074/jbc.RA119.009954. Epub 2019 Jul 30. J Biol Chem. 2019. PMID: 31362986 Free PMC article.
-
Rapid desensitization of humanized mice with anti-human FcεRIα monoclonal antibodies.J Allergy Clin Immunol. 2020 Mar;145(3):907-921.e3. doi: 10.1016/j.jaci.2019.12.003. Epub 2019 Dec 11. J Allergy Clin Immunol. 2020. PMID: 31836406 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

