Intramuscular versus intravenous oxytocin to prevent postpartum haemorrhage at vaginal delivery: randomised controlled trial
- PMID: 30181338
- PMCID: PMC6122278
- DOI: 10.1136/bmj.k3546
Intramuscular versus intravenous oxytocin to prevent postpartum haemorrhage at vaginal delivery: randomised controlled trial
Abstract
Objective: To determine whether intravenous oxytocin is more effective than intramuscular oxytocin at preventing postpartum haemorrhage at vaginal delivery.
Design: Double blind placebo controlled randomised trial.
Setting: University affiliated maternity unit in the Republic of Ireland.
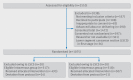
Participants: 1075 women aged 18 years or older, at term with a singleton pregnancy who were aiming for a vaginal delivery with an actively managed third stage of labour.
Interventions: Women were allocated to an intravenous bolus of oxytocin (10 IU in 1 mL given slowly over one minute) and placebo intramuscular injection (1 mL 0.9% saline) or an intramuscular bolus of oxytocin (10 IU in 1 mL) and placebo intravenous injection (1 mL 0.9% saline given slowly over one minute) at vaginal delivery. Allocation was by a secure web based randomisation service with masking of participants and clinicians to the trial intervention.
Main outcome measures: The primary outcome was postpartum haemorrhage (PPH, measured blood loss ≥500 mL). Secondary outcomes were severe PPH (measured blood loss ≥1000 mL), need for blood transfusion, admission to a high dependency unit, and side effects to oxytocin.
Results: Between 4 January 2016 and 13 December 2017, 1075 women were randomised and 1035 (96.3%) included in the primary and secondary analyses (517 in the intravenous oxytocin group and 518 in the intramuscular oxytocin group). The incidence of PPH was not significantly lower in the intravenous group (18.8%, 97/517) compared with intramuscular group (23.2%, 120/518): adjusted odds ratio 0.75 (95% confidence interval 0.55 to 1.03). The incidence of severe PPH, however, was significantly lower in the intravenous group (4.6%, 24/517) compared with intramuscular group (8.1%, 42/518): 0.54 (0.32 to 0.91) as was the need for blood transfusion (1.5% v 4.4%, 0.31, 0.13 to 0.70) and admission to a high dependency unit (1.7% v 3.7%, 0.44, 0.20 to 0.98). The number needed to treat to prevent one case of severe PPH was 29 (95% confidence interval 16 to 201) and to prevent one case of blood transfusion was 35 (20 to 121). The incidence of side effects to oxytocin was not increased in the intravenous group compared with intramuscular group (4.1% v 5.2%, 0.75, 0.42 to 1.35).
Conclusion: Intravenous oxytocin for the third stage of labour results in less frequent severe PPH, blood transfusion, and admission to a high dependency unit than intramuscular oxytocin, and without excess side effects.
Trial registration: Current Controlled Trials ISRCTN14718882.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://group.bmj.com/group/rights-licensing/permissions.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at http://www.icmje.org/coi_disclosure.pdf (available on request from the corresponding author) and declare: no support from any organisation for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
References
-
- Department of Reproductive Health and Research WHO WHO Recommendations for the prevention and treatment of postpartum haemorrhage. World Health Organization, 2012. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical