Activation of pro-survival metabolic networks by 1,25(OH)2D3 does not hamper the sensitivity of breast cancer cells to chemotherapeutics
- PMID: 30181873
- PMCID: PMC6116450
- DOI: 10.1186/s40170-018-0183-6
Activation of pro-survival metabolic networks by 1,25(OH)2D3 does not hamper the sensitivity of breast cancer cells to chemotherapeutics
Abstract
Background: We have previously identified 1,25-dihydroxyvitamin D3 [1,25(OH)2D3], the bioactive form of vitamin D3, as a potent regulator of energy-utilization and nutrient-sensing pathways in prostate cancer cells. In the current study, we investigated the effects of 1,25(OH)2D3 on breast cancer (BCa) cell metabolism using cell lines representing distinct molecular subtypes, luminal (MCF-7 and T-47D), and triple-negative BCa (MDA-MB-231, MDA-MB-468, and HCC-1143).
Methods: 1,25(OH)2D3's effect on BCa cell metabolism was evaluated by employing a combination of real-time measurements of glycolysis/oxygen consumption rates using a biosensor chip system, GC/MS-based metabolomics, gene expression analysis, and assessment of overall energy levels. The influence of treatment on energy-related signaling molecules was investigated by immunoblotting.
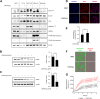
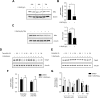
Results: We show that 1,25(OH)2D3 significantly induces the expression and activity of the pentose phosphate pathway enzyme glucose-6-phosphate dehydrogenase (G6PD) in all BCa cell lines, however differentially influences glycolytic and respiratory rates in the same cells. Although 1,25(OH)2D3 treatment was found to induce seemingly anti-oxidant responses in MCF-7 cells, such as increased intracellular serine levels, and reduce the expression of its putative target gene thioredoxin-interacting protein (TXNIP), intracellular reactive oxygen species levels were found to be elevated. Serine accumulation in 1,25(OH)2D3-treated cells was not found to hamper the efficacy of chemotherapeutics, including 5-fluorouracil. Detailed analyses of the nature of TXNIP's regulation by 1,25(OH)2D3 included genetic and pharmacological inhibition of signaling molecules and metabolic enzymes including AMP-activated protein kinase and G6PD, as well as by studying the ITCH (E3 ubiquitin ligase)-TXNIP interaction. While these investigations demonstrated minimal involvement of such pathways in the observed non-canonical regulation of TXNIP, inhibition of estrogen receptor (ER) signaling by tamoxifen mirrored the reduction of TXNIP levels by 1,25(OH)2D3, demonstrating that the latter's negative regulation of ER expression is a potential mechanism of TXNIP modulation.
Conclusions: Altogether, we propose that regulation of energy metabolism contributes to 1,25(OH)2D3's anti-cancer effects and that combining 1,25(OH)2D3 with drugs targeting metabolic networks in tumor cells may lead to synergistic effects.
Keywords: AMPK; Breast cancer; Estrogen receptor; G6PD; ITCH; Metabolism; Serine; TXNIP; Vitamin D.
Conflict of interest statement
Not applicableNot applicableThe authors declare that they have no competing interests.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures




References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

