Cytotoxic effects of heated tobacco products (HTP) on human bronchial epithelial cells
- PMID: 30185530
- PMCID: PMC6252481
- DOI: 10.1136/tobaccocontrol-2018-054317
Cytotoxic effects of heated tobacco products (HTP) on human bronchial epithelial cells
Abstract
Background: Heated tobacco product(s) (HTP), also called heat-not-burn products, are a re-emerging class of tobacco products that purport to reduce health risk compared with smoking combustible tobacco products. This study examined the potential toxic effects of inhaling emissions from an HTP in comparison with electronic and combustible tobacco cigarettes.
Methods: Inhalation toxicity of HTP (IQOS; tobacco flavour), e-cigarette (MarkTen; tobacco flavour) and tobacco cigarette (Marlboro Red) was examined in vitro using an air-liquid interface with human bronchial epithelial cells (H292). Cells were exposed directly to 55 puffs from the e-cigarette, 12 puffs from the HTP and 8 puffs from the tobacco cigarette to equilibrate nicotine delivery to the cells across products. Cytotoxicity was measured using neutral red uptake and trypan blue assays. Cytotoxic effects of each tested product (HTP, e-cigarette and tobacco cigarette) were compared with an air control. Release of inflammatory markers (cytokines) was measured using ELISA.
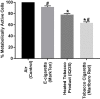
Results: The HTP showed higher cytotoxicity compared with the air controls using the neutral red assay. The HTP also showed higher cytotoxicity than the e-cigarette, but lower cytotoxicity than the combustible cigarettes using the same assay. A significant increase in cytokines levels, compared with air controls, was observed postexposure to tobacco smoke but not to emissions from HTP or e-cigarette aerosol.
Discussion: Using limited cytotoxic measures, the HTP showed reduced cytotoxicity relative to a combustible cigarette but higher toxicity than an e-cigarette. More comprehensive testing is needed to determine long-term effects of inhaling emissions from HTP.
Keywords: nicotine; non-cigarette tobacco products; toxicology.
© Author(s) (or their employer(s)) 2018. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: MLG reports grants from and served as an advisory board member to pharmaceutical companies that manufacture smoking cessation drugs. RJO was a member of the FDA Tobacco Products Scientific Advisory Committee which considered Philip Morris International’s modified risk application for IQOS in January 2018. Other authors declare no conflict of interest.
Figures
Similar articles
-
Characterisation of the borgwaldt LM4E system for in vitro exposures to undiluted aerosols from next generation tobacco and nicotine products (NGPs).Food Chem Toxicol. 2018 Mar;113:337-344. doi: 10.1016/j.fct.2018.02.005. Epub 2018 Feb 5. Food Chem Toxicol. 2018. PMID: 29421647
-
Comparison of the chemical composition of aerosols from heated tobacco products, electronic cigarettes and tobacco cigarettes and their toxic impacts on the human bronchial epithelial BEAS-2B cells.J Hazard Mater. 2021 Jan 5;401:123417. doi: 10.1016/j.jhazmat.2020.123417. Epub 2020 Jul 7. J Hazard Mater. 2021. PMID: 32763707
-
Concurrent Choice Assessment of Preference and Substitutability of E-cigarettes and Heated Tobacco Products for Combustible Cigarettes Among African American and White Smokers.Nicotine Tob Res. 2023 Jul 14;25(8):1505-1508. doi: 10.1093/ntr/ntad052. Nicotine Tob Res. 2023. PMID: 37042345 Free PMC article.
-
Heated Tobacco Products: Insights into Composition and Toxicity.Toxics. 2023 Aug 2;11(8):667. doi: 10.3390/toxics11080667. Toxics. 2023. PMID: 37624172 Free PMC article. Review.
-
New ideas, old problems? Heated tobacco products - a systematic review.Int J Occup Med Environ Health. 2019 Oct 16;32(5):595-634. doi: 10.13075/ijomeh.1896.01433. Epub 2019 Sep 26. Int J Occup Med Environ Health. 2019. PMID: 31584041
Cited by
-
Insight into the pulmonary molecular toxicity of heated tobacco products using human bronchial and alveolar mucosa models at air-liquid interface.Sci Rep. 2022 Sep 30;12(1):16396. doi: 10.1038/s41598-022-20657-y. Sci Rep. 2022. PMID: 36180488 Free PMC article.
-
Awareness of heated tobacco products among US Adults - Health information national trends survey, 2020.Subst Abus. 2022;43(1):1023-1034. doi: 10.1080/08897077.2022.2060440. Subst Abus. 2022. PMID: 35435808 Free PMC article.
-
A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Survival and Growth of Common Oral Commensal Streptococci.Int J Environ Res Public Health. 2019 May 14;16(10):1669. doi: 10.3390/ijerph16101669. Int J Environ Res Public Health. 2019. PMID: 31091650 Free PMC article.
-
Pathophysiological Responses of Oral Keratinocytes After Exposure to Flavored E-Cigarette Liquids.Dent J (Basel). 2025 Jan 29;13(2):60. doi: 10.3390/dj13020060. Dent J (Basel). 2025. PMID: 39996934 Free PMC article.
-
Association between exposure to secondhand aerosol from heated tobacco products and respiratory symptoms among current non-smokers in Japan: a cross-sectional study.BMJ Open. 2023 Mar 7;13(3):e065322. doi: 10.1136/bmjopen-2022-065322. BMJ Open. 2023. PMID: 36882244 Free PMC article.
References
-
- Zanetti F, Titz B, Sewer A, et al. . Comparative systems toxicology analysis of cigarette smoke and aerosol from a candidate modified risk tobacco product in organotypic human gingival epithelial cultures: a 3-day repeated exposure study. Food Chem Toxicol 2017;101:15–35. 10.1016/j.fct.2016.12.027 - DOI - PubMed
-
- Wong ET, Kogel U, Veljkovic E, et al. . Evaluation of the Tobacco Heating System 2.2. Part 4: 90-day OECD 413 rat inhalation study with systems toxicology endpoints demonstrates reduced exposure effects compared with cigarette smoke. Regul Toxicol Pharmacol 2016;81:S59–S81. 10.1016/j.yrtph.2016.10.015 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
