Important Shapeshifter: Mechanisms Allowing Astrocytes to Respond to the Changing Nervous System During Development, Injury and Disease
- PMID: 30186118
- PMCID: PMC6111612
- DOI: 10.3389/fncel.2018.00261
Important Shapeshifter: Mechanisms Allowing Astrocytes to Respond to the Changing Nervous System During Development, Injury and Disease
Abstract
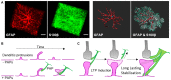
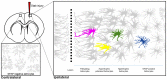
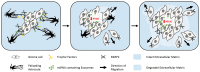
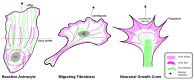
Astrocytes are the most prevalent glial cells in the brain. Historically considered as "merely supporting" neurons, recent research has shown that astrocytes actively participate in a large variety of central nervous system (CNS) functions including synaptogenesis, neuronal transmission and synaptic plasticity. During disease and injury, astrocytes efficiently protect neurons by various means, notably by sealing them off from neurotoxic factors and repairing the blood-brain barrier. Their ramified morphology allows them to perform diverse tasks by interacting with synapses, blood vessels and other glial cells. In this review article, we provide an overview of how astrocytes acquire their complex morphology during development. We then move from the developing to the mature brain, and review current research on perisynaptic astrocytic processes, with a particular focus on how astrocytes engage synapses and modulate their formation and activity. Comprehensive changes have been reported in astrocyte cell shape in many CNS pathologies. Factors influencing these morphological changes are summarized in the context of brain pathologies, such as traumatic injury and degenerative conditions. We provide insight into the molecular, cellular and cytoskeletal machinery behind these shape changes which drive the dynamic remodeling in astrocyte morphology during injury and the development of pathologies.
Keywords: CNS; astrocytes; astrogliosis; brain trauma; cytoskeleton; morphology; pathology; synapse.
Figures


Similar articles
-
Astrocyte morphology: Diversity, plasticity, and role in neurological diseases.CNS Neurosci Ther. 2019 Jun;25(6):665-673. doi: 10.1111/cns.13123. Epub 2019 Mar 30. CNS Neurosci Ther. 2019. PMID: 30929313 Free PMC article. Review.
-
Imaging and Manipulating Astrocyte Function In Vivo in the Context of CNS Injury.Methods Mol Biol. 2019;1938:233-246. doi: 10.1007/978-1-4939-9068-9_16. Methods Mol Biol. 2019. PMID: 30617984
-
Astrocyte-synapse interactions and cell adhesion molecules.FEBS J. 2023 Jul;290(14):3512-3526. doi: 10.1111/febs.16540. Epub 2022 Jun 19. FEBS J. 2023. PMID: 35647709 Review.
-
Role of astrocytes in synapse formation and maturation.Curr Top Dev Biol. 2021;142:371-407. doi: 10.1016/bs.ctdb.2020.12.010. Epub 2021 Jan 21. Curr Top Dev Biol. 2021. PMID: 33706922 Review.
-
Perisynaptic astroglial processes: dynamic processors of neuronal information.Brain Struct Funct. 2016 Jun;221(5):2427-42. doi: 10.1007/s00429-015-1070-3. Epub 2015 May 31. Brain Struct Funct. 2016. PMID: 26026482 Review.
Cited by
-
Inflammation Drives Alzheimer's Disease: Emphasis on 5-lipoxygenase Pathways.Curr Neuropharmacol. 2021;19(6):885-895. doi: 10.2174/1570159X18666200924122732. Curr Neuropharmacol. 2021. PMID: 32972344 Free PMC article. Review.
-
Automated detection of GFAP-labeled astrocytes in micrographs using YOLOv5.Sci Rep. 2022 Dec 23;12(1):22263. doi: 10.1038/s41598-022-26698-7. Sci Rep. 2022. PMID: 36564441 Free PMC article.
-
Ketamine Alters Functional Plasticity of Astroglia: An Implication for Antidepressant Effect.Life (Basel). 2021 Jun 17;11(6):573. doi: 10.3390/life11060573. Life (Basel). 2021. PMID: 34204579 Free PMC article. Review.
-
Therapeutic activities of naringenin on efavirenz-induced sleep-like disorder in the midbrain of white albino mice.Iran J Basic Med Sci. 2020 Nov;23(11):1462-1470. doi: 10.22038/ijbms.2020.47043.10852. Iran J Basic Med Sci. 2020. PMID: 33235704 Free PMC article.
-
IFI204 in microglia mediates traumatic brain injury-induced mitochondrial dysfunction and pyroptosis via SENP7 interaction.Cell Biol Toxicol. 2025 May 23;41(1):89. doi: 10.1007/s10565-025-10032-8. Cell Biol Toxicol. 2025. PMID: 40407969 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

