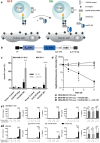
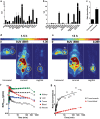
Development of a novel target module redirecting UniCAR T cells to Sialyl Tn-expressing tumor cells
- PMID: 30190468
- PMCID: PMC6127150
- DOI: 10.1038/s41408-018-0113-4
Development of a novel target module redirecting UniCAR T cells to Sialyl Tn-expressing tumor cells
Conflict of interest statement
L.R.L., C.N., M.B., and P.V. have filed patents related to anti-STn mAb L2A5. M.B. has filed patents related to the UniCAR system. M.B. is shareholder of the company GEMoaB which owns the IP related to the UniCAR system. The remaining authors declare that they have no conflict of interest.
Figures
References
-
- Sadelain M, Brentjens R, Rivière I. The basic principles of chimeric antigen receptor design. Cancer Discov. 2013;3:388–98. doi: 10.1158/2159-8290.CD-12-0548. - DOI - PMC - PubMed
-
- Kochenderfer JN, Dudley ME, Feldman SA, Wilson WH, Spaner DE, Maric I, et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood. 2012;119:2709–2720. doi: 10.1182/blood-2011-10-384388. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

