Farnesylated heat shock protein 40 is a component of membrane-bound RISC in Arabidopsis
- PMID: 30194279
- PMCID: PMC6204899
- DOI: 10.1074/jbc.RA118.003887
Farnesylated heat shock protein 40 is a component of membrane-bound RISC in Arabidopsis
Abstract
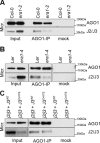
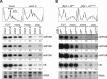
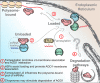
ARGONAUTE1 (AGO1) binds directly to small regulatory RNA and is a key effector protein of post-transcriptional gene silencing mediated by microRNA (miRNA) and small interfering RNA (siRNA) in Arabidopsis The formation of an RNA-induced silencing complex (RISC) of AGO1 and small RNA requires the function of the heat shock protein 70/90 chaperone system. Some functions of AGO1 occur in association with endomembranes, in particular the rough endoplasmic reticulum (RER), but proteins interacting with AGO1 in membrane fractions remain unidentified. In this study, we show that the farnesylated heat shock protein 40 homologs, J2 and J3, associate with AGO1 in membrane fractions in a manner that involves protein farnesylation. We also show that three changes in AGO1 function are detectable in mutants in protein farnesylation and J2/J3. First, perturbations of the HSP40/70/90 pathway by mutation of J3, HSP90, and farnesyl transferase affect the amounts of AGO1 associated with membranes. Second, miRNA association with membrane-bound polysomes is increased in farnesyl transferase and farnesylation-deficient J2/J3 mutants. Third, silencing by noncell autonomously acting short interfering RNAs is impaired. These observations highlight the involvement of farnesylated J2/J3 in small RNA-mediated gene regulation, and suggest that the importance of chaperone-AGO1 interaction is not limited to the RISC assembly process.
Keywords: ARGONAUTE; Arabidopsis thaliana; Argonaute; Chaperones; Protein farnesylation; Rough Endoplasmic Reticulum; Small RNA; chaperone DnaJ (DnaJ); membrane; microRNA mechanism.
© 2018 Sjögren et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures


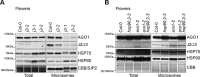



References
-
- Rivas F. V., Tolia N. H., Song J. J., Aragon J. P., Liu J., Hannon G. J., and Joshua-Tor L. (2005) Purified Argonaute2 and an siRNA form recombinant human RISC. Nat. Struct. Mol. Biol. 12, 340–349 - PubMed
-
- Iwasaki S., Kobayashi M., Yoda M., Sakaguchi Y., Katsuma S., Suzuki T., and Tomari Y. (2010) Hsc70/Hsp90 chaperone machinery mediates ATP-dependent RISC loading of small RNA duplexes. Mol. Cell 39, 292–299 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

