Influence of Dlutaraldehyde Cross-Linking Modes on the Recyclability of Immobilized Lipase B from Candida antarctica for Transesterification of Soy Bean Oil
- PMID: 30200521
- PMCID: PMC6225267
- DOI: 10.3390/molecules23092230
Influence of Dlutaraldehyde Cross-Linking Modes on the Recyclability of Immobilized Lipase B from Candida antarctica for Transesterification of Soy Bean Oil
Abstract
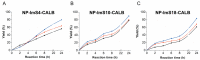
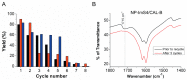
Lipase B from Candida antarctica (CAL-B) is largely employed as a biocatalyst for hydrolysis, esterification, and transesterification reactions. CAL-B is a good model enzyme to study factors affecting the enzymatic structure, activity and/or stability after an immobilization process. In this study, we analyzed the immobilization of CAL-B enzyme on different magnetic nanoparticles, synthesized by the coprecipitation method inside inverse micelles made of zwitterionic surfactants, with distinct carbon chain length: 4 (ImS4), 10 (ImS10) and 18 (ImS18) carbons. Magnetic nanoparticles ImS4 and ImS10 were shown to cross-link to CAL-B enzyme via a Michael-type addition, whereas particles with ImS18 were bond via pyridine formation after glutaraldehyde cross-coupling. Interestingly, the Michael-type cross-linking generated less stable immobilized CAL-B, revealing the influence of a cross-linking mode on the resulting biocatalyst behavior. Curiously, a direct correlation between nanoparticle agglomerate sizes and CAL-B enzyme reuse stability was observed. Moreover, free CAL-B enzyme was not able to catalyze transesterification due to the high methanol concentration; however, the immobilized CAL-B enzyme reached yields from 79.7 to 90% at the same conditions. In addition, the transesterification of lipids isolated from oleaginous yeasts achieved 89% yield, which confirmed the potential of immobilized CAL-B enzyme in microbial production of biodiesel.
Keywords: biodiesel synthesis; cross-linking types; enzyme immobilization; lipase; magnetic nanoparticles.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
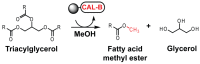

Similar articles
-
Magnetic Cross-Linked Enzyme Aggregates (mCLEAs) of Candida antarctica lipase: an efficient and stable biocatalyst for biodiesel synthesis.PLoS One. 2014 Dec 31;9(12):e115202. doi: 10.1371/journal.pone.0115202. eCollection 2014. PLoS One. 2014. PMID: 25551445 Free PMC article.
-
Lipase-immobilized biocatalytic membranes for biodiesel production.Bioresour Technol. 2013 Oct;145:229-32. doi: 10.1016/j.biortech.2012.12.054. Epub 2012 Dec 19. Bioresour Technol. 2013. PMID: 23357586
-
Immobilization of Candida antarctica Lipase B on Magnetic Poly(Urea-Urethane) Nanoparticles.Appl Biochem Biotechnol. 2016 Oct;180(3):558-575. doi: 10.1007/s12010-016-2116-6. Epub 2016 May 16. Appl Biochem Biotechnol. 2016. PMID: 27184256
-
Biodiesel production with immobilized lipase: A review.Biotechnol Adv. 2010 Sep-Oct;28(5):628-34. doi: 10.1016/j.biotechadv.2010.05.012. Epub 2010 May 24. Biotechnol Adv. 2010. PMID: 20580809 Review.
-
Biodiesel production by transesterification using immobilized lipase.Biotechnol Lett. 2013 Apr;35(4):479-90. doi: 10.1007/s10529-012-1116-z. Epub 2012 Dec 18. Biotechnol Lett. 2013. PMID: 23247566 Review.
Cited by
-
Microbial Lipases and Their Potential in the Production of Pharmaceutical Building Blocks.Int J Mol Sci. 2022 Sep 1;23(17):9933. doi: 10.3390/ijms23179933. Int J Mol Sci. 2022. PMID: 36077332 Free PMC article. Review.
-
Bioreceptor modified electrochemical biosensors for the detection of life threating pathogenic bacteria: a review.RSC Adv. 2024 Sep 6;14(39):28487-28515. doi: 10.1039/d4ra04038d. eCollection 2024 Sep 4. RSC Adv. 2024. PMID: 39247512 Free PMC article. Review.
-
Enhanced Biodiesel Production with Eversa Transform 2.0 Lipase on Magnetic Nanoparticles.Langmuir. 2024 Dec 24;40(51):26835-26851. doi: 10.1021/acs.langmuir.4c02542. Epub 2024 Nov 26. Langmuir. 2024. PMID: 39591534 Free PMC article.
-
Molecular Decoration of Ceramic Supports for Highly Effective Enzyme Immobilization-Material Approach.Materials (Basel). 2021 Jan 3;14(1):201. doi: 10.3390/ma14010201. Materials (Basel). 2021. PMID: 33401646 Free PMC article.
-
The use of scanning electron microscopy and fixation methods to evaluate the interaction of blood with the surfaces of medical devices.Sci Rep. 2024 Feb 26;14(1):4622. doi: 10.1038/s41598-024-55136-z. Sci Rep. 2024. PMID: 38409219 Free PMC article.
References
-
- Sirisha V.L., Jain A., Jain A. Enzyme immobilization: An overview on methods, support material, and applications of immobilized enzymes. Adv. Food Nutr. Res. 2016;79:179–211. - PubMed
-
- Netto C.G.C.M., Toma H.E., Andrade L.H. Superparamagnetic nanoparticles as versatile carriers and supporting materials for enzymes. J. Mol. Catal. B-Enzym. 2013;85–86:71–92. doi: 10.1016/j.molcatb.2012.08.010. - DOI
-
- Zdarta J., Meyer A.S., Jesionowski T., Pinelo M. A general overview of support materials for enzyme immobilization: Characteristics, properties, practical utility. Catalysts. 2018;8:92. doi: 10.3390/catal8020092. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

