Engineering the fatty acid synthesis pathway in Synechococcus elongatus PCC 7942 improves omega-3 fatty acid production
- PMID: 30202434
- PMCID: PMC6123915
- DOI: 10.1186/s13068-018-1243-4
Engineering the fatty acid synthesis pathway in Synechococcus elongatus PCC 7942 improves omega-3 fatty acid production
Abstract
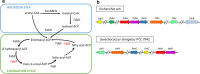
Background: The microbial production of fatty acids has received great attention in the last few years as feedstock for the production of renewable energy. The main advantage of using cyanobacteria over other organisms is their ability to capture energy from sunlight and to transform CO2 into products of interest by photosynthesis, such as fatty acids. Fatty acid synthesis is a ubiquitous and well-characterized pathway in most bacteria. However, the activity of the enzymes involved in this pathway in cyanobacteria remains poorly explored.
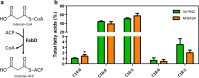
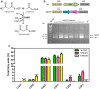
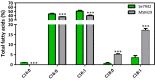
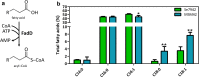
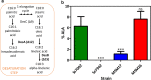
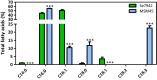
Results: To characterize the function of some enzymes involved in the saturated fatty acid synthesis in cyanobacteria, we genetically engineered Synechococcus elongatus PCC 7942 by overexpressing or deleting genes encoding enzymes of the fatty acid synthase system and tested the lipid profile of the mutants. These modifications were in turn used to improve alpha-linolenic acid production in this cyanobacterium. The mutant resulting from fabF overexpression and fadD deletion, combined with the overexpression of desA and desB desaturase genes from Synechococcus sp. PCC 7002, produced the highest levels of this omega-3 fatty acid.
Conclusions: The fatty acid composition of S. elongatus PCC 7942 can be significantly modified by genetically engineering the expression of genes coding for the enzymes involved in the first reactions of fatty acid synthesis pathway. Variations in fatty acid composition of S. elongatus PCC 7942 mutants did not follow the pattern observed in Escherichia coli derivatives. Some of these modifications can be used to improve omega-3 fatty acid production. This work provides new insights into the saturated fatty acid synthesis pathway and new strategies that might be used to manipulate the fatty acid content of cyanobacteria.
Keywords: Cyanobacteria; Fatty acid synthesis; Omega-3 fatty acids; Synechococcus elongatus PCC 7942; fab genes.
Figures

Similar articles
-
Highlighting the potential of Synechococcus elongatus PCC 7942 as platform to produce α-linolenic acid through an updated genome-scale metabolic modeling.Front Microbiol. 2023 Mar 14;14:1126030. doi: 10.3389/fmicb.2023.1126030. eCollection 2023. Front Microbiol. 2023. PMID: 36998399 Free PMC article.
-
Synechococcus elongatus PCC 7942 as a Platform for Bioproduction of Omega-3 Fatty Acids.Life (Basel). 2022 May 29;12(6):810. doi: 10.3390/life12060810. Life (Basel). 2022. PMID: 35743841 Free PMC article.
-
Cloning of omega 3 desaturase from cyanobacteria and its use in altering the degree of membrane-lipid unsaturation.Plant Mol Biol. 1994 Oct;26(1):249-63. doi: 10.1007/BF00039536. Plant Mol Biol. 1994. PMID: 7524725
-
Improved Free Fatty Acid Production in Cyanobacteria with Synechococcus sp. PCC 7002 as Host.Front Bioeng Biotechnol. 2014 May 26;2:17. doi: 10.3389/fbioe.2014.00017. eCollection 2014. Front Bioeng Biotechnol. 2014. PMID: 25152890 Free PMC article.
-
Photosynthetic symbiotic therapeutics - An innovative, effective treatment for ischemic cardiovascular diseases.J Mol Cell Cardiol. 2022 Mar;164:51-57. doi: 10.1016/j.yjmcc.2021.11.007. Epub 2021 Nov 20. J Mol Cell Cardiol. 2022. PMID: 34813842 Review.
Cited by
-
Cyanobacteria as a Promising Alternative for Sustainable Environment: Synthesis of Biofuel and Biodegradable Plastics.Front Microbiol. 2022 Jul 13;13:939347. doi: 10.3389/fmicb.2022.939347. eCollection 2022. Front Microbiol. 2022. PMID: 35903468 Free PMC article. Review.
-
LFQRatio: A Normalization Method to Decipher Quantitative Proteome Changes in Microbial Coculture Systems.J Proteome Res. 2024 Mar 1;23(3):999-1013. doi: 10.1021/acs.jproteome.3c00714. Epub 2024 Feb 14. J Proteome Res. 2024. PMID: 38354288 Free PMC article.
-
Acyl-lipid desaturases and Vipp1 cooperate in cyanobacteria to produce novel omega-3 PUFA-containing glycolipids.Biotechnol Biofuels. 2020 May 6;13:83. doi: 10.1186/s13068-020-01719-7. eCollection 2020. Biotechnol Biofuels. 2020. PMID: 32399061 Free PMC article.
-
Enhanced Lipid Production and Molecular Dynamics under Salinity Stress in Green Microalga Chlamydomonas reinhardtii (137C).Mar Drugs. 2019 Aug 20;17(8):484. doi: 10.3390/md17080484. Mar Drugs. 2019. PMID: 31434347 Free PMC article.
-
Cyanobacterial circadian regulation enhances bioproduction under subjective nighttime through rewiring of carbon partitioning dynamics, redox balance orchestration, and cell cycle modulation.Microb Cell Fact. 2025 Mar 8;24(1):56. doi: 10.1186/s12934-025-02665-5. Microb Cell Fact. 2025. PMID: 40055679 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

