Elobixibat for the treatment of constipation
- PMID: 30204504
- PMCID: PMC6386599
- DOI: 10.1080/17474124.2018.1522248
Elobixibat for the treatment of constipation
Abstract
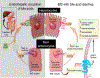
Chronic idiopathic constipation (CC) is highly prevalent worldwide. A subset of patients with CC have reduced fecal (and by inference, intra-colonic) bile acids (BA). Elobixibat, a locally-acting ileal bile acid transporter (IBAT) inhibitor, leads to increased BA delivery to the colon and represents a new class of treatment for CC. BAs accelerate colonic transit and increase colonic secretion. Therefore, IBAT inhibitors have potential to treat patients with CC. Areas covered: Rationale for IBAT inhibitor in therapeutics, and preclinical and clinical pharmacology of elobixibat: In vitro, elobixibat is a highly potent, selective IBAT inhibitor. In humans, elobixibat accelerated colonic transit. In phase 2A, 2B and 3 studies in CC, elobixibat was efficacious, well tolerated and safe. An open-label, phase 3 trial (52 weeks) confirmed the safety of elobixibat. Elobixibat reduces LDL cholesterol, increases serum GLP-1, and has potential in metabolic syndrome. Expert commentary: Uniquely among current treatments of CC, elobixibat stimulates both motor and secretory functions in the colon. These dual effects suggest that, when approved, elobixibat may be a first-line choice for constipation associated with colonic BA deficiency and a second-line treatment for all patients with CC and constipation-predominant irritable bowel syndrome. Further studies are required to confirm efficacy for relief of CC. Once approved, elobixibat will likely become a second-line choice for treatment of CC.
Keywords: Bile acid; enterohepatic circulation; ileal bile acid transporter (IBAT); irritable bowel syndrome; pharmacodynamics; pharmacokinetics.
Figures


References
-
- Shin A, Camilleri M, Vijayvargiya P, Busciglio I, Burton D, Ryks M, Rhoten D, Lueke A, Saenger A, Girtman A, Zinsmeister AR: Bowel functions, fecal unconjugated primary and secondary bile acids, and colonic transit in patients with irritable bowel syndrome. Clin Gastroenterol Hepatol (2013) 11(10):1270–1275 e1271. - PMC - PubMed
-
- Wedlake L, A’Hern R, Russell D, Thomas K, Walters JR, Andreyev HJ: Systematic review: The prevalence of idiopathic bile acid malabsorption as diagnosed by sehcat scanning in patients with diarrhoea-predominant irritable bowel syndrome. Alimentary pharmacology & therapeutics (2009) 30(7):707–717. - PubMed
-
- Valentin N, Camilleri M, Altayar O, Vijayvargiya P, Acosta A, Nelson AD, Murad MH. Biomarkers for bile acid diarrhea in functional bowel disorder with diarrhea: a systematic review and meta-analysis. Gut (2016) 65(12):1951–1959. - PubMed
-
- Vijayvargiya P, Camilleri M: Update on bile acid malabsorption: Finally ready for prime time? Current Gastroenterology Reports (2018) 20(3):10. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
