Iron catalysis of lipid peroxidation in ferroptosis: Regulated enzymatic or random free radical reaction?
- PMID: 30217775
- PMCID: PMC6555767
- DOI: 10.1016/j.freeradbiomed.2018.09.008
Iron catalysis of lipid peroxidation in ferroptosis: Regulated enzymatic or random free radical reaction?
Abstract
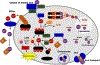
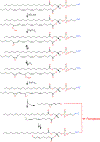
Duality of iron as an essential cofactor of many enzymatic metabolic processes and as a catalyst of poorly controlled redox-cycling reactions defines its possible biological beneficial and hazardous role in the body. In this review, we discuss these two "faces" of iron in a newly conceptualized program of regulated cell death, ferroptosis. Ferroptosis is a genetically programmed iron-dependent form of regulated cell death driven by enhanced lipid peroxidation and insufficient capacity of thiol-dependent mechanisms (glutathione peroxidase 4, GPX4) to eliminate hydroperoxy-lipids. We present arguments favoring the enzymatic mechanisms of ferroptotically engaged non-heme iron of 15-lipoxygenases (15-LOX) in complexes with phosphatidylethanolamine binding protein 1 (PEBP1) as a catalyst of highly selective and specific oxidation reactions of arachidonoyl- (AA) and adrenoyl-phosphatidylethanolamines (PE). We discuss possible role of iron chaperons as control mechanisms for guided iron delivery directly to their "protein clients" thus limiting non-enzymatic redox-cycling reactions. We also consider opportunities of loosely-bound iron to contribute to the production of pro-ferroptotic lipid oxidation products. Finally, we propose a two-stage iron-dependent mechanism for iron in ferroptosis by combining its catalytic role in the 15-LOX-driven production of 15-hydroperoxy-AA-PE (HOO-AA-PE) as well as possible involvement of loosely-bound iron in oxidative cleavage of HOO-AA-PE to oxidatively truncated electrophiles capable of attacking nucleophilic targets in yet to be identified proteins leading to cell demise.
Keywords: 15-lipoxygenase; Ferroptosis; GPX4; Glutathione; Hydroperoxy-arachidonoyl-phosphatidylethanolamine; Iron; Iron chaperons; Lipid peroxidation.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures


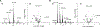
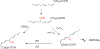
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 CA165065/CA/NCI NIH HHS/United States
- R01 ES020693/ES/NIEHS NIH HHS/United States
- U19 AI068021/AI/NIAID NIH HHS/United States
- R37 NS061817/NS/NINDS NIH HHS/United States
- R01 NS076511/NS/NINDS NIH HHS/United States
- R01 HL136143/HL/NHLBI NIH HHS/United States
- P30 DA035778/DA/NIDA NIH HHS/United States
- P01 HL114453/HL/NHLBI NIH HHS/United States
- R01 GM113908/GM/NIGMS NIH HHS/United States
- R01 HL086884/HL/NHLBI NIH HHS/United States
- R01 NS061817/NS/NINDS NIH HHS/United States
- P41 GM103712/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

