Aspirin promotes apoptosis and inhibits proliferation by blocking G0/G1 into S phase in rheumatoid arthritis fibroblast-like synoviocytes via downregulation of JAK/STAT3 and NF-κB signaling pathway
- PMID: 30221683
- PMCID: PMC6202076
- DOI: 10.3892/ijmm.2018.3883
Aspirin promotes apoptosis and inhibits proliferation by blocking G0/G1 into S phase in rheumatoid arthritis fibroblast-like synoviocytes via downregulation of JAK/STAT3 and NF-κB signaling pathway
Abstract
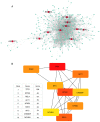
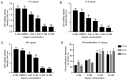
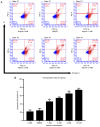
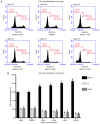
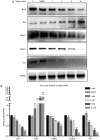
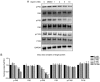
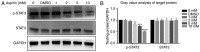
Rheumatoid arthritis (RA) is a commonly occurring autoimmune disease. Its defining pathological characteristic is the excessive proliferation of fibroblast‑like synoviocytes (FLS), which is similar to tumor cells and results in a range of clinical problems. As a commonly used antipyretic, analgesic and anti‑inflammatory drug, aspirin is the first‑line treatment for RA. However, its mechanism of action has not been well explained. The goal is to investigate the biological effects of aspirin on primary RA‑FLS and its underlying mechanisms. In this experiment we treated cells with various concentrations of aspirin (0, DMSO, 1, 2, 5, 10 mM). Cell proliferation activity was detected with CCK‑8 assays. Apoptosis and cell cycle distribution were detected via flow cytometry. Apoptosis and cell cycle‑associated proteins (Bcl‑2, Bax, PRAP1, Cyclin D1, P21), as well as the key proteins and their phosphorylation levels of the NF‑κB and JAK/STAT3 signaling pathways, were detected via western blot analysis. Bioinformatics prediction revealed that aspirin was closely associated with cell proliferation and apoptosis, including the p53 and NF‑κB signaling pathways. By stimulating with aspirin, cell viability decreased, while the proportion of apoptotic cells increased, and the number of cells arrested in the G0/G1 phase increased in a dose‑dependent manner. The expression of Bax increased with aspirin stimulation, while the levels of Bcl‑2, PRAP1, Cyclin D1 and P21 decreased; p‑STAT3, p‑P65 and p‑50 levels also decreased while STAT3, P65, P50, p‑P105 and P105 remained unchanged. From our data, it can be concluded that aspirin is able to promote apoptosis and inhibit the proliferation of RA‑FLS through blocking the JAK/STAT3 and NF‑κB signaling pathways.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

