Myocardial insufficiency is related to reduced subunit 4 content of cytochrome c oxidase
- PMID: 30223867
- PMCID: PMC6142347
- DOI: 10.1186/s13019-018-0785-7
Myocardial insufficiency is related to reduced subunit 4 content of cytochrome c oxidase
Abstract
Background: Treatment of heart failure remains one of the most challenging task for intensive care medicine, cardiology and cardiac surgery. New options and better indicators are always required. Understanding the basic mechanisms underlying heart failure promote the development of adjusted therapy e.g. assist devices and monitoring of recovery. If cardiac failure is related to compromised cellular respiration of the heart, remains unclear. Myocardial respiration depends on Cytochrome c- Oxidase (CytOx) activity representing the rate limiting step for the mitochondrial respiratory chain. The enzymatic activity as well as mRNA expression of enzyme's mitochondrial encoded catalytic subunit 2, nuclear encoded regulatory subunit 4 and protein contents were studied in biopsies of cardiac patients suffering from myocardial insufficiency and dilated cardiomyopathy (DCM).
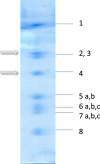
Methods: Fifty-four patients were enrolled in the study and underwent coronary angiography. Thirty male patients (mean age: 45 +/- 15 yrs.) had a reduced ejection fraction (EF) 35 ± 12% below 45% and a left ventricular end diastolic diameter (LVEDD) of 71 ± 10 mm bigger than 56 mm. They were diagnosed as having idiopathic dilated cardiomyopathy (DCM) without coronary heart disease and NYHA-class 3 and 4. Additionally, 24 male patients (mean age: 52 +/- 11 yrs.) after exclusion of secondary cardiomyopathies, coronary artery or valve disease, served as control (EF: 68 ± 7, LVEDD: 51 ± 7 mm). Total RNA was extracted from two biopsies of each person. Real-time PCR analysis was performed with specific primers followed by a melt curve analysis. Corresponding protein expression in the tissue was studied with immune-histochemistry while enzymatic activity was evaluated by spectroscopy.
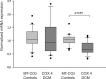
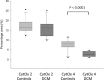
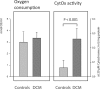
Results: Gene and protein expression analysis of patients showed a significant decrease of subunit 4 (1.1 vs. 0.6, p < 0.001; 7.7 ± 3.1% vs. 2.8 ± 1.4%, p < 0.0001) but no differences in subunit 2. Correlations were found between reduced subunit 2 expression, low EF (r = 0.766, p < 0.00045) and increased LVEDD (r = 0.492, p < 0.0068). In case of DCM less subunit 4 expression and reduced shortening fraction (r = 0.524, p < 0.017) was found, but enzymatic activity was higher (0.08 ± 0.06 vs. 0.26 ± 0.08 U/mg, p < 0.001) although myocardial oxygen consumption continued to the same extent.
Conclusion: In case of myocardial insufficiency and DCM, decreased expression of COX 4 results in an impaired CytOx activity. Higher enzymatic activity but equal oxygen consumption contribute to the pathophysiology of the myocardial insufficiency and appears as an indicator of oxidative stress. This kind of dysregulation should be in the focus for the development of diagnostic and therapy procedures.
Keywords: Cytochrome c oxidase; MT –CO2 and COX 4 expression; Myocardial insufficiency.
Conflict of interest statement
Ethical approval and consent to participate
The Ethics Committee at the University Hospital of Gießen and Marburg (UKGM) has approved the study (AZ: 53/18). All patients gave their informed consent to use leftover samples for scientific purposes.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Effects of surgical ventricular reconstruction and mitral complex reconstruction on cardiac oxidative metabolism and efficiency in nonischemic and ischemic dilated cardiomyopathy.JACC Cardiovasc Imaging. 2011 Jul;4(7):762-70. doi: 10.1016/j.jcmg.2011.04.010. JACC Cardiovasc Imaging. 2011. PMID: 21757167
-
Alterations in myocardial tissue factor expression and cellular localization in dilated cardiomyopathy.J Am Coll Cardiol. 2005 Apr 5;45(7):1081-9. doi: 10.1016/j.jacc.2004.12.061. J Am Coll Cardiol. 2005. PMID: 15808768
-
Mitochondrial active and relaxed state respiration after heat shock mRNA response in the heart.J Therm Biol. 2019 Feb;80:106-112. doi: 10.1016/j.jtherbio.2019.01.007. Epub 2019 Jan 9. J Therm Biol. 2019. PMID: 30784473
-
Mechanisms of toxic cardiomyopathy.Clin Toxicol (Phila). 2019 Jan;57(1):1-9. doi: 10.1080/15563650.2018.1497172. Epub 2018 Sep 27. Clin Toxicol (Phila). 2019. PMID: 30260248 Review.
-
Mitochondrial respiration is controlled by Allostery, Subunit Composition and Phosphorylation Sites of Cytochrome c Oxidase: A trailblazer's tale - Bernhard Kadenbach.Mitochondrion. 2021 Sep;60:228-233. doi: 10.1016/j.mito.2021.08.015. Epub 2021 Sep 2. Mitochondrion. 2021. PMID: 34481964 Review.
Cited by
-
An elemental diet protects mouse salivary glands from 5-fluorouracil-induced atrophy.Oncol Lett. 2022 Jun;23(6):178. doi: 10.3892/ol.2022.13298. Epub 2022 Apr 14. Oncol Lett. 2022. PMID: 35464303 Free PMC article.
-
Role of Trientine in Hypertrophic Cardiomyopathy: A Review of Mechanistic Aspects.Pharmaceuticals (Basel). 2022 Sep 14;15(9):1145. doi: 10.3390/ph15091145. Pharmaceuticals (Basel). 2022. PMID: 36145368 Free PMC article. Review.
-
Cost-Effective Fiber Optic Solutions for Biosensing.Biosensors (Basel). 2022 Jul 28;12(8):575. doi: 10.3390/bios12080575. Biosensors (Basel). 2022. PMID: 36004971 Free PMC article. Review.
-
Proteomic and Global DNA Methylation Modulation in Lipid Metabolism Disorders with a Marine-Derived Bioproduct.Biology (Basel). 2023 Jun 2;12(6):806. doi: 10.3390/biology12060806. Biology (Basel). 2023. PMID: 37372091 Free PMC article.
-
Mapping drug-target interactions and synergy in multi-molecular therapeutics for pressure-overload cardiac hypertrophy.NPJ Syst Biol Appl. 2021 Feb 15;7(1):11. doi: 10.1038/s41540-021-00171-z. NPJ Syst Biol Appl. 2021. PMID: 33589646 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

