Early toxicity and patient reported quality-of-life in patients receiving proton therapy for localized prostate cancer: a single institutional review of prospectively recorded outcomes
- PMID: 30223877
- PMCID: PMC6142310
- DOI: 10.1186/s13014-018-1127-6
Early toxicity and patient reported quality-of-life in patients receiving proton therapy for localized prostate cancer: a single institutional review of prospectively recorded outcomes
Abstract
Background: We report prospectively captured clinical toxicity and patient reported outcomes in a single institutional cohort of patients treated for prostate cancer with proton beam therapy (PBT). This is the largest reported series of patients treated mostly with pencil beam scanning PBT.
Methods: We reviewed 231 patients treated on an IRB approved institutional registry from 2013 to 2016; final analysis included 192 patients with > 1-year of follow-up. Toxicity incidence was prospectively captured and scored using CTCAE v4.0. International Prostate Symptoms Score (IPSS), Sexual Health Inventory for Men (SHIM) score, and Expanded Prostate Cancer Index Composite (EPIC) bowel domain questionnaires were collected at each visit. Univariate Cox regression was used to explore associations of grade 2+ toxicity with clinical, treatment, and dosimetric variables.
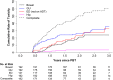
Results: Median follow-up was 1.7 years. Grade 3 toxicity was seen in 5/192 patients. No grade 4 or 5 toxicity was seen. Patient reported quality-of-life showed no change in urinary function post-radiation by IPSS scores. Median SHIM scores declined by 3.7 points at 1-year post-treatment without further decrease beyond year 1. On univariate analysis, only younger age (HR = 0.61, p = 0.022) was associated with decreased sexual toxicity. EPIC bowel domain scores declined from 96 at baseline (median) by an average of 5.4 points at 1-year post-treatment (95% CI: 2.5-8.2 points, p < 0.001), with no further decrease over time. Bowel toxicity was mostly in the form of transient rectal bleeding and was associated with anticoagulation use (HR = 3.45, p = 0.002).
Conclusions: Grade 3 or higher toxicity was rare at 2-years after treatment with PBT for localized prostate cancer. Longer follow-up is needed to further characterize late toxicity and biochemical control.
Trial registration: NCT, NCT01255748 . Registered 1 January 2013.
Keywords: Patient reported outcomes; Prostate cancer; Proton therapy; Quality of life.
Conflict of interest statement
Ethics approval and consent to participate
All patients were enrolled on a prospective University of Washington IRB approved registry and gave written consent for their participation.
Consent for publication
Not applicable. All patient information has been thoroughly de-identified.
Competing interests
Mr. Hippe reports grants from the NIH/NCI, and Dr. Russell reports a scientific advisory position with Varian Medical Corporation during the conduct of this study. All remaining authors of this publication declare no competing interest.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

Similar articles
-
Five-Year Biochemical Results, Toxicity, and Patient-Reported Quality of Life After Delivery of Dose-Escalated Image Guided Proton Therapy for Prostate Cancer.Int J Radiat Oncol Biol Phys. 2016 May 1;95(1):422-434. doi: 10.1016/j.ijrobp.2016.02.038. Epub 2016 Feb 16. Int J Radiat Oncol Biol Phys. 2016. PMID: 27084658 Clinical Trial.
-
Rectal Hydrogel Spacer Improves Late Gastrointestinal Toxicity Compared to Rectal Balloon Immobilization After Proton Beam Radiation Therapy for Localized Prostate Cancer: A Retrospective Observational Study.Int J Radiat Oncol Biol Phys. 2020 Nov 1;108(3):635-643. doi: 10.1016/j.ijrobp.2020.01.026. Epub 2020 Feb 6. Int J Radiat Oncol Biol Phys. 2020. PMID: 32035187
-
Four-Year Outcomes From a Prospective Phase II Clinical Trial of Moderately Hypofractionated Proton Therapy for Localized Prostate Cancer.Int J Radiat Oncol Biol Phys. 2019 Nov 15;105(4):713-722. doi: 10.1016/j.ijrobp.2019.05.069. Epub 2019 Jun 11. Int J Radiat Oncol Biol Phys. 2019. PMID: 31199994 Clinical Trial.
-
Proton therapy for prostate cancer: A review of the rationale, evidence, and current state.Urol Oncol. 2019 Sep;37(9):628-636. doi: 10.1016/j.urolonc.2018.11.012. Epub 2018 Dec 4. Urol Oncol. 2019. PMID: 30527342 Review.
-
The Use of Particle Therapy in Prostate Cancer.Semin Radiat Oncol. 2025 Jul;35(3):353-361. doi: 10.1016/j.semradonc.2025.03.001. Semin Radiat Oncol. 2025. PMID: 40516970 Review.
Cited by
-
Acute toxicity and patient-reported symptom score after conventional versus moderately hypofractionated proton therapy for prostate cancer.J Med Radiat Sci. 2022 Jun;69(2):198-207. doi: 10.1002/jmrs.551. Epub 2021 Oct 19. J Med Radiat Sci. 2022. PMID: 34664410 Free PMC article.
-
Prostate Cancer Treatment with Pencil Beam Proton Therapy Using Rectal Spacers sans Endorectal Balloons.Int J Part Ther. 2022 Apr 6;9(1):28-41. doi: 10.14338/IJPT-21-00039. eCollection 2022 Summer. Int J Part Ther. 2022. PMID: 35774493 Free PMC article.
-
Long-Term Patient-Reported Bowel and Urinary Quality of Life in Patients Treated with Intensity-Modulated Radiotherapy Versus Intensity-Modulated Proton Therapy for Localized Prostate Cancer.Curr Oncol. 2025 Apr 2;32(4):212. doi: 10.3390/curroncol32040212. Curr Oncol. 2025. PMID: 40277768 Free PMC article.
References
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

