Intranasal Corticosteroids Do Not Lead to Ocular Changes: A Systematic Review and Meta-analysis
- PMID: 30229924
- PMCID: PMC6320292
- DOI: 10.1002/lary.27209
Intranasal Corticosteroids Do Not Lead to Ocular Changes: A Systematic Review and Meta-analysis
Abstract
Objectives: The safety and efficacy of intranasal corticosteroids (INCS) are well established, but there remains apprehension that INCS could lead to systemic side effects, as with oral steroids. The objective of this systematic review was to assess whether the use of INCS lead to increased intraocular pressure (IOP) above 20 mm Hg, glaucoma, or formation of posterior subcapsular cataracts in adult patients with rhinitis.
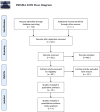
Methods: Two medical librarians searched the published literature for records discussing the use of "nasal steroids" in "rhinitis" and their effect on "intraocular pressure," "cataracts," or "glaucoma."
Results: A total of 484 studies were identified, and 10 randomized controlled trials met our inclusion criteria. Meta-analysis of 2,226 patients revealed that the relative risk of elevated IOP in those who received INCS was 2.24 (95% confidence interval [CI]: 0.68 to 7.34) compared to placebo. The absolute increased incidence of elevated IOP in patients using INCS compared to placebo was 0.8% (95% CI: 0% to 1.6%). There were zero cases of glaucoma in both placebo and INCS groups at 12 months. The absolute increased incidence of developing a posterior subcapsular cataract was 0.02% (95% CI: -0.3% to 0.4%).
Conclusions: Use of INCS is not associated with a significant risk of elevating IOP or developing a posterior subcapsular cataract in patients with allergic rhinitis. Presence of glaucoma, however, is the real clinical adverse event of concern. There were zero reported cases of glaucoma at 12 months. Future studies should formally evaluate for glaucoma rather than use IOP measures as a surrogate. Laryngoscope, 129:6-12, 2019.
Keywords: Intranasal steroids; cataract formation; glaucoma; intraocular pressure.
© 2018 The American Laryngological, Rhinological and Otological Society, Inc.
Conflict of interest statement
The authors have no other funding, financial relationships, or conflicts of interest to disclose.
Figures


References
-
- Seidman MD, Gurgel RK, Lin SY, et al. Clinical practice guideline: Allergic rhinitis. Otolaryngol Head Neck Surg. 2015;152:S1–43. - PubMed
-
- Bousquet J, Van Cauwenberge P, Khaltaev N, Aria Workshop G, World Health O Allergic rhinitis and its impact on asthma. J Allergy Clin Immunol. 2001;108:S147–334. - PubMed
-
- Bascom R, Wachs M, Naclerio RM, Pipkorn U, Galli SJ, Lichtenstein LM. Basophil influx occurs after nasal antigen challenge: effects of topical corticosteroid pretreatment. J Allergy Clin Immunol. 1988;81:580–589. - PubMed
-
- Bisgaard H, Gronborg H, Mygind N, Dahl R, Lindqvist N, Venge P. Allergen-induced increase of eosinophil cationic protein in nasal lavage fluid: effect of the glucocorticoid budesonide. J Allergy Clin Immunol. 1990;85:891–895. - PubMed
-
- Bradding P, Feather IH, Wilson S, Holgate ST, Howarth PH. Cytokine immunoreactivity in seasonal rhinitis: regulation by a topical corticosteroid. Am J Respir Crit Care Med. 1995;151:1900–1906. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

