Muscle does not drive persistent posttraumatic elbow contracture in a rat model
- PMID: 30230560
- PMCID: PMC6296879
- DOI: 10.1002/mus.26344
Muscle does not drive persistent posttraumatic elbow contracture in a rat model
Abstract
Introduction: Posttraumatic elbow contracture is clinically challenging because injury often disrupts multiple periarticular soft tissues. Tissue specific contribution to contracture, particularly muscle, remains poorly understood.
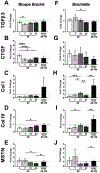
Methods: In this study we used a previously developed animal model of elbow contracture. After surgically inducing a unilateral soft tissue injury, injured limbs were immobilized for 3, 7, 21, and 42 days (IM) or for 42 IM with 42 days of free mobilization (42/42 IM-FM). Biceps brachii active/passive mechanics and morphology were evaluated at 42 IM and 42/42 IM-FM, whereas biceps brachii and brachialis gene expression was evaluated at all time points.
Results: Injured limb muscle exhibited significantly altered active/passive mechanics and decreased fiber area at 42 IM but returned to control levels by 42/42 IM-FM. Gene expression suggested muscle growth rather than a fibrotic response at 42/42 IM-FM.
Discussion: Muscle is a transient contributor to motion loss in our rat model of posttraumatic elbow contracture. Muscle Nerve 58:843-851, 2018.
Keywords: contracture; elbow; fibrosis; gene expression; mechanics; muscle.
© 2018 Wiley Periodicals, Inc.
Conflict of interest statement
Figures
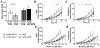
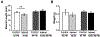


Similar articles
-
The Role of Periarticular Soft Tissues in Persistent Motion Loss in a Rat Model of Posttraumatic Elbow Contracture.J Bone Joint Surg Am. 2019 Mar 6;101(5):e17. doi: 10.2106/JBJS.18.00246. J Bone Joint Surg Am. 2019. PMID: 30845041 Free PMC article.
-
Temporal Patterns of Motion in Flexion-extension and Pronation-supination in a Rat Model of Posttraumatic Elbow Contracture.Clin Orthop Relat Res. 2018 Sep;476(9):1878-1889. doi: 10.1097/CORR.0000000000000388. Clin Orthop Relat Res. 2018. PMID: 30001292 Free PMC article.
-
Persistent motion loss after free joint mobilization in a rat model of post-traumatic elbow contracture.J Shoulder Elbow Surg. 2017 Apr;26(4):611-618. doi: 10.1016/j.jse.2016.09.059. Epub 2017 Jan 10. J Shoulder Elbow Surg. 2017. PMID: 28081997 Free PMC article.
-
Perspectives on glenohumeral joint contractures and shoulder dysfunction in children with perinatal brachial plexus palsy.J Hand Ther. 2015 Apr-Jun;28(2):176-83; quiz 184. doi: 10.1016/j.jht.2014.12.001. Epub 2014 Dec 18. J Hand Ther. 2015. PMID: 25835253 Review.
-
Posttraumatic elbow contractures: targeting neuroinflammatory fibrogenic mechanisms.J Orthop Sci. 2013 Nov;18(6):869-77. doi: 10.1007/s00776-013-0447-5. Epub 2013 Sep 5. J Orthop Sci. 2013. PMID: 24005582 Review.
Cited by
-
Early Joint Use Following Elbow Dislocation Limits Range-of-Motion Loss and Tissue Pathology in Posttraumatic Joint Contracture.J Bone Joint Surg Am. 2023 Feb 1;105(3):223-230. doi: 10.2106/JBJS.22.00064. Epub 2022 Dec 2. J Bone Joint Surg Am. 2023. PMID: 36723466 Free PMC article.
-
Ultrasound Pressure-Dependent Cytokine and Immune Cell Response Lost in Aged Muscle.Ultrasound Med Biol. 2024 Apr;50(4):494-501. doi: 10.1016/j.ultrasmedbio.2023.12.009. Epub 2024 Jan 12. Ultrasound Med Biol. 2024. PMID: 38218683 Free PMC article.
-
Pleiotropic Effects of Simvastatin and Losartan in Preclinical Models of Post-Traumatic Elbow Contracture.Front Bioeng Biotechnol. 2022 Feb 21;10:803403. doi: 10.3389/fbioe.2022.803403. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35265595 Free PMC article.
-
Preclinical Models of Elbow Injury and Pathology.Ann Jt. 2021 Jan;6:12. doi: 10.21037/aoj.2020.02.09. Epub 2021 Jan 15. Ann Jt. 2021. PMID: 35990575 Free PMC article.
-
The pronator contracture syndrome: A new entity in supination restriction.Shoulder Elbow. 2025 Jan;17(1):96-103. doi: 10.1177/17585732241239031. Epub 2024 Mar 22. Shoulder Elbow. 2025. PMID: 39552670 Free PMC article.
References
-
- Trudel G, Uhthoff HK. Contractures secondary to immobility: is the restriction articular or muscular? An experimental longitudinal study in the rat knee. Arch Phys Med Rehabil 2000;81(1):6–13. - PubMed
-
- King GJ, Morrey BF, An K-N. Stabilizers of the elbow. JSES 1993;2(3):165–174. - PubMed
-
- Duckworth AD, Kulijdian A, McKee MD, Ring D. Residual subluxation of the elbow after dislocation or fracture-dislocation: treatment with active elbow exercises and avoidance of varus stress. JSES 2008;17(2):276–280. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

