Direct Transamidation Reactions: Mechanism and Recent Advances
- PMID: 30231486
- PMCID: PMC6225162
- DOI: 10.3390/molecules23092382
Direct Transamidation Reactions: Mechanism and Recent Advances
Abstract
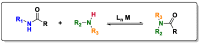
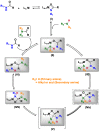
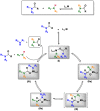
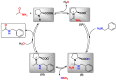
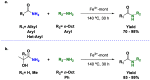
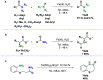
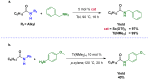
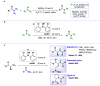
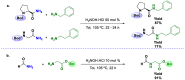
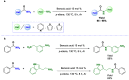
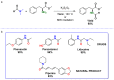
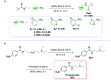
Amides are undeniably some of the most important compounds in Nature and the chemical industry, being present in biomolecules, materials, pharmaceuticals and many other substances. Unfortunately, the traditional synthesis of amides suffers from some important drawbacks, principally the use of stoichiometric activators or the need to use highly reactive carboxylic acid derivatives. In recent years, the transamidation reaction has emerged as a valuable alternative to prepare amides. The reactivity of amides makes their direct reaction with nitrogen nucleophiles difficult; thus, the direct transamidation reaction needs a catalyst in order to activate the amide moiety and to promote the completion of the reaction because equilibrium is established. In this review, we present research on direct transamidation reactions ranging from studies of the mechanism to the recent developments of more applicable and versatile methodologies, emphasizing those reactions involving activation with metal catalysts.
Keywords: amide; amine; catalysis; catalyst; transamidation.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures



Similar articles
-
Catalytic transamidation reactions compatible with tertiary amide metathesis under ambient conditions.J Am Chem Soc. 2009 Jul 29;131(29):10003-8. doi: 10.1021/ja8094262. J Am Chem Soc. 2009. PMID: 19621957
-
A CO2-Catalyzed Transamidation Reaction.J Org Chem. 2021 Dec 3;86(23):16867-16881. doi: 10.1021/acs.joc.1c02077. Epub 2021 Nov 1. J Org Chem. 2021. PMID: 34723529
-
Catalytic transamidation under moderate conditions.J Am Chem Soc. 2003 Mar 26;125(12):3422-3. doi: 10.1021/ja028242h. J Am Chem Soc. 2003. PMID: 12643691
-
Direct Access to Amides from Nitro-Compounds via Aminocarbonylation and Amidation Reactions: A Minireview.Chem Rec. 2021 Dec;21(12):4059-4087. doi: 10.1002/tcr.202100224. Epub 2021 Sep 2. Chem Rec. 2021. PMID: 34472167 Review.
-
Recent Advances in the Metal-Catalyzed Activation of Amide Bonds.Chem Asian J. 2019 Jan 4;14(1):76-93. doi: 10.1002/asia.201801317. Epub 2018 Dec 6. Chem Asian J. 2019. PMID: 30426696 Review.
Cited by
-
Base-promoted dynamic amide exchange: efficient access to isotopically enriched tertiary amides.Chem Sci. 2025 Jul 15;16(34):15528-36. doi: 10.1039/d5sc02566d. Online ahead of print. Chem Sci. 2025. PMID: 40747321 Free PMC article.
-
Innovative application of magnetically modified bovine horn as a natural keratin resource in the role of value-added organocatalyst.RSC Adv. 2022 Jun 6;12(26):16535-16543. doi: 10.1039/d1ra09327d. eCollection 2022 Jun 1. RSC Adv. 2022. PMID: 35754867 Free PMC article.
-
Transamidation of Amides and Amidation of Esters by Selective N-C(O)/O-C(O) Cleavage Mediated by Air- and Moisture-Stable Half-Sandwich Nickel(II)-NHC Complexes.Molecules. 2021 Jan 2;26(1):188. doi: 10.3390/molecules26010188. Molecules. 2021. PMID: 33401664 Free PMC article.
-
Advances in Metal-Free Transamidation: A Sustainable Approach to Amide Bond Formation.Chem Asian J. 2025 Jun;20(12):e202500327. doi: 10.1002/asia.202500327. Epub 2025 Apr 10. Chem Asian J. 2025. PMID: 40084501 Free PMC article. Review.
-
C-Terminal Arginine-Selective Cleavage of Peptides as a Method for Mimicking Carboxypeptidase B.Org Lett. 2023 Aug 25;25(33):6206-6210. doi: 10.1021/acs.orglett.3c02418. Epub 2023 Aug 16. Org Lett. 2023. PMID: 37585337 Free PMC article.
References
-
- Greenberg A., Breneman C.M., Liebman J.F. The Amide Linkage: Structural Significance in Chemistry, Biochemistry, and Materials Science. Wiley-Interscience; New York, NY, USA: 2000.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

