Biodegradation of ECM hydrogel promotes endogenous brain tissue restoration in a rat model of stroke
- PMID: 30232030
- PMCID: PMC6217851
- DOI: 10.1016/j.actbio.2018.09.020
Biodegradation of ECM hydrogel promotes endogenous brain tissue restoration in a rat model of stroke
Abstract
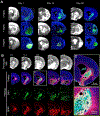
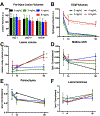
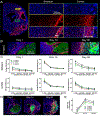
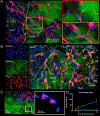
The brain is considered to have a limited capacity to repair damaged tissue and no regenerative capacity following injury. Tissue lost after a stroke is therefore not spontaneously replaced. Extracellular matrix (ECM)-based hydrogels implanted into the stroke cavity can attract endogenous cells. These hydrogels can be formulated at different protein concentrations that govern their rheological and inductive properties. We evaluated histologically 0, 3, 4 and 8 mg/mL of porcine-derived urinary bladder matrix (UBM)-ECM hydrogel concentrations implanted in a 14-day old stroke cavity. Less concentrated hydrogels (3 and 4 mg/mL) were efficiently degraded with a 95% decrease in volume by 90 days, whereas only 32% of the more concentrated and stiffer hydrogel (8 mg/mL) was resorbed. Macrophage infiltration and density within the bioscaffold progressively increased in the less concentrated hydrogels and decreased in the 8 mg/mL hydrogels. The less concentrated hydrogels showed a robust invasion of endothelial cells with neovascularization. No neovascularization occurred with the stiffer hydrogel. Invasion of neural cells increased with time in all hydrogel concentrations. Differentiation of neural progenitors into mature neurons with axonal projections was evident, as well as a robust invasion of oligodendrocytes. However, relatively few astrocytes were present in the ECM hydrogel, although some were present in the newly forming tissue between degrading scaffold patches. Implantation of an ECM hydrogel partially induced neural tissue restoration, but a more complete understanding is required to evaluate its potential therapeutic application. STATEMENT OF SIGNIFICANCE: Extracellular matrix hydrogel promotes tissue regeneration in many peripheral soft tissues. However, the brain has generally been considered to lack the potential for tissue regeneration. We here demonstrate that tissue regeneration in the brain can be achieved using implantation of ECM hydrogel into a tissue cavity. A structure-function relationship is key to promote tissue regeneration in the brain. Specifically, weaker hydrogels that were retained in the cavity underwent an efficient biodegradation within 14 days post-implantation to promote a tissue restoration within the lesion cavity. In contrast, stiffer ECM hydrogel only underwent minor biodegradation and did not lead to a tissue restoration. Inductive hydrogels weaker than brain tissue provide the appropriate condition to promote an endogenous regenerative response that restores tissue in a cavity. This approach offers new avenues for the future treatment of chronic tissue damage caused by stroke and other acute brain injuries.
Keywords: Biodegradation; Biomaterial; Cell invasion; Extracellular matrix; Hydrogel; Magnetic resonance imaging; Regeneration; Scaffold; Stroke; Tissue repair.
Copyright © 2018 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
Figures






References
-
- Baddour JA, Sousounis K, Tsonis PA, Organ repair and regeneration: an overview, Birth Defects Res C Embryo Today 96(1) (2012) 1–29. - PubMed
-
- Michalopoulos GK, DeFrances MC, Liver regeneration, Science 276(5309) (1997) 60–6. - PubMed
-
- Bechmann I, Failed central nervous system regeneration: a downside of immune privilege?, Neuromolecular Med 7(3) (2005) 217–28. - PubMed
-
- Tanaka EM, Ferretti P, Considering the evolution of regeneration in the central nervous system, Nat Rev Neurosci 10(10) (2009) 713–23. - PubMed
-
- Fry EJ, Central nervous system regeneration: mission impossible?, Clin Exp Pharmacol Physiol 28(4) (2001) 253–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

