Subcellular Targeting of Theranostic Radionuclides
- PMID: 30233374
- PMCID: PMC6131480
- DOI: 10.3389/fphar.2018.00996
Subcellular Targeting of Theranostic Radionuclides
Abstract
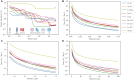
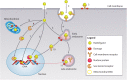
The last decade has seen rapid growth in the use of theranostic radionuclides for the treatment and imaging of a wide range of cancers. Radionuclide therapy and imaging rely on a radiolabeled vector to specifically target cancer cells. Radionuclides that emit β particles have thus far dominated the field of targeted radionuclide therapy (TRT), mainly because the longer range (μm-mm track length) of these particles offsets the heterogeneous expression of the molecular target. Shorter range (nm-μm track length) α- and Auger electron (AE)-emitting radionuclides on the other hand provide high ionization densities at the site of decay which could overcome much of the toxicity associated with β-emitters. Given that there is a growing body of evidence that other sensitive sites besides the DNA, such as the cell membrane and mitochondria, could be critical targets in TRT, improved techniques in detecting the subcellular distribution of these radionuclides are necessary, especially since many β-emitting radionuclides also emit AE. The successful development of TRT agents capable of homing to targets with subcellular precision demands the parallel development of quantitative assays for evaluation of spatial distribution of radionuclides in the nm-μm range. In this review, the status of research directed at subcellular targeting of radionuclide theranostics and the methods for imaging and quantification of radionuclide localization at the nanoscale are described.
Keywords: cancer; dosimetry; radioimmunotherapy; radiopharmaceuticals; subcellular targeting; targeted radionuclide therapy.
Figures



References
-
- Alberts B., Johnson A., Lewis J., Raff M., Roberts K., Walter P. (2002). “Fractionation of cells,” in Molecular Biology of the Cell, ed. Drubin D. (New York, NY: Garland Sciences; ).
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

