Global Disruption of α2A Adrenoceptor Barely Affects Bone Tissue but Minimizes the Detrimental Effects of Thyrotoxicosis on Cortical Bone
- PMID: 30233491
- PMCID: PMC6127616
- DOI: 10.3389/fendo.2018.00486
Global Disruption of α2A Adrenoceptor Barely Affects Bone Tissue but Minimizes the Detrimental Effects of Thyrotoxicosis on Cortical Bone
Abstract
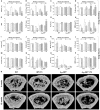
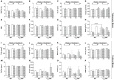
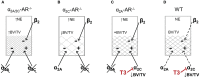
Evidence shows that sympathetic nervous system (SNS) activation inhibits bone formation and activates bone resorption leading to bone loss. Because thyroid hormone (TH) interacts with the SNS to control several physiological processes, we raised the hypothesis that this interaction also controls bone remodeling. We have previously shown that mice with double-gene inactivation of α2A- and -adrenoceptors (α2A/2C-AR-/-) present high bone mass (HBM) phenotype and resistance to thyrotoxicosis-induced osteopenia, which supports a TH-SNS interaction to control bone mass and suggests that it involves α2-AR signaling. Accordingly, we detected expression of α2A-AR, α2B-AR and α2C-AR in the skeleton, and that triiodothyronine (T3) modulates α2C-AR mRNA expression in the bone. Later, we found that mice with single-gene inactivation of α2C-AR (α2C-AR-/-) present low bone mass in the femur and HBM in the vertebra, but that both skeletal sites are resistant to TH-induce osteopenia, showing that the SNS actions occur in a skeletal site-dependent manner, and that thyrotoxicosis depends on α2C-AR signaling to promote bone loss. To further dissect the specific roles of α2-AR subtypes, in this study, we evaluated the skeletal phenotype of mice with single-gene inactivation of α2A-AR (α2A-AR-/-), and the effect of daily treatment with a supraphysiological dose of T3, for 4 or 12 weeks, on bone microarchitecture and bone resistance to fracture. Micro-computed tomographic (μCT) analysis revealed normal trabecular and cortical bone structure in the femur and vertebra of euthyroid α2A-AR-/- mice. Thyrotoxicosis was more detrimental to femoral trabecular bone in α2A-AR-/- than in WT mice, whereas this bone compartment had been previously shown to present resistance to thyrotoxicosis in α2C-AR-/- mice. Altogether these findings reveal that TH excess depends on α2C-AR signaling to negatively affect femoral trabecular bone. In contrast, thyrotoxicosis was more deleterious to femoral and vertebral cortical bone in WT than in α2A-AR-/- mice, suggesting that α2A-AR signaling contributes to TH actions on cortical bone. These findings further support a TH-SNS interaction to control bone physiology, and suggest that α2A-AR and α2C-AR signaling pathways have key roles in the mechanisms through which thyrotoxicosis promotes its detrimental effects on bone remodeling, structure and resistance to fracture.
Keywords: bone remodeling; cortical bone; sympathetic nervous system; thyroid hormone; thyrotoxicosis; trabecular bone; α2A-adrenoceptor.
Figures
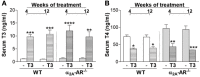
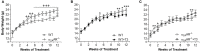
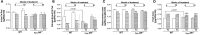
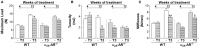

Similar articles
-
Lack of α2C-Adrenoceptor Results in Contrasting Phenotypes of Long Bones and Vertebra and Prevents the Thyrotoxicosis-Induced Osteopenia.PLoS One. 2016 Jan 27;11(1):e0146795. doi: 10.1371/journal.pone.0146795. eCollection 2016. PLoS One. 2016. PMID: 26815679 Free PMC article.
-
Thyrotoxicosis Involves β2-Adrenoceptor Signaling to Negatively Affect Microarchitecture and Biomechanical Properties of the Femur.Thyroid. 2019 Aug;29(8):1060-1072. doi: 10.1089/thy.2018.0259. Thyroid. 2019. PMID: 31264512
-
Double disruption of α2A- and α2C-adrenoceptors results in sympathetic hyperactivity and high-bone-mass phenotype.J Bone Miner Res. 2011 Mar;26(3):591-603. doi: 10.1002/jbmr.243. J Bone Miner Res. 2011. PMID: 20814988
-
Therapeutic Potential of Selectively Targeting the α2C-Adrenoceptor in Cognition, Depression, and Schizophrenia-New Developments and Future Perspective.Front Psychiatry. 2017 Aug 14;8:144. doi: 10.3389/fpsyt.2017.00144. eCollection 2017. Front Psychiatry. 2017. PMID: 28855875 Free PMC article. Review.
-
α2C-adrenoceptor modulators: a patent review.Expert Opin Ther Pat. 2011 Apr;21(4):455-81. doi: 10.1517/13543776.2011.565333. Expert Opin Ther Pat. 2011. PMID: 21413828 Review.
Cited by
-
Degradation products of magnesium implant synergistically enhance bone regeneration: Unraveling the roles of hydrogen gas and alkaline environment.Bioact Mater. 2024 Dec 26;46:331-346. doi: 10.1016/j.bioactmat.2024.12.020. eCollection 2025 Apr. Bioact Mater. 2024. PMID: 39816475 Free PMC article.
References
-
- Varga F, Rumpler M, Luegmayr E, Fratzl-Zelman N, Glantschnig H, Klaushofer K. Triiodothyronine, a regulator of osteoblastic differentiation: depression of histone H4, attenuation of c-fos/c-jun, and induction of osteocalcin expression. Calcif Tissue Int. (1997) 61:404–11. 10.1007/s002239900356 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

