BABA-Primed Histone Modifications in Potato for Intergenerational Resistance to Phytophthora infestans
- PMID: 30233606
- PMCID: PMC6135045
- DOI: 10.3389/fpls.2018.01228
BABA-Primed Histone Modifications in Potato for Intergenerational Resistance to Phytophthora infestans
Abstract
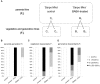
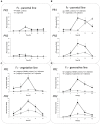
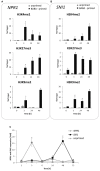
In this paper we analyzed β-aminobutyric acid (BABA)-primed epigenetic adjustment of potato cv. "Sarpo Mira" to Phytophthora infestans. The first stress-free generation of the potato genotype obtained from BABA-primed parent plants via tubers and seeds showed pronounced resistance to the pathogen, which was tuned with the transcriptional memory of SA-responsive genes. During the early priming phase before the triggering stress, we found robust bistable deposition of histone marks (H3K4me2 and H3K27me3) on the NPR1 (Non-expressor of PR genes) and the SNI1 gene (Suppressor of NPR1, Inducible), in which transcription antagonized silencing. Switchable chromatin states of these adverse systemic acquired resistance (SAR) regulators probably reprogrammed responsiveness of the PR1 and PR2 genes and contributed to stress imprinting. The elevated levels of heritable H3K4me2 tag in the absence of transcription on SA-dependent genes in BABA-primed (F0) and its vegetative and generative progeny (F1) before pathogen challenge provided evidence for the epigenetic mark for intergenerational memory in potato. Moreover, our study revealed that histone acetylation was not critical for maintaining BABA-primed defense information until the plants were triggered with the virulent pathogen when rapid and boosted PRs gene expression probably required histone acetyltransferase (HAT) activity both in F0 and F1 progeny.
Keywords: histone modifications; intergenerational SAR; late blight; potato leaves; priming.
Figures

Similar articles
-
BABA-primed defense responses to Phytophthora infestans in the next vegetative progeny of potato.Front Plant Sci. 2015 Oct 15;6:844. doi: 10.3389/fpls.2015.00844. eCollection 2015. Front Plant Sci. 2015. PMID: 26528308 Free PMC article.
-
BABA-Induced DNA Methylome Adjustment to Intergenerational Defense Priming in Potato to Phytophthora infestans.Front Plant Sci. 2019 May 31;10:650. doi: 10.3389/fpls.2019.00650. eCollection 2019. Front Plant Sci. 2019. PMID: 31214209 Free PMC article.
-
Normoergic NO-dependent changes, triggered by a SAR inducer in potato, create more potent defense responses to Phytophthora infestans.Plant Sci. 2013 Oct;211:23-34. doi: 10.1016/j.plantsci.2013.06.007. Epub 2013 Jun 21. Plant Sci. 2013. PMID: 23987808
-
Role of NPR1 and KYP in long-lasting induced resistance by β-aminobutyric acid.Front Plant Sci. 2014 May 8;5:184. doi: 10.3389/fpls.2014.00184. eCollection 2014. Front Plant Sci. 2014. PMID: 24847342 Free PMC article.
-
Comparative cDNA-AFLP analysis reveals that DL-beta-amino-butyric acid induces resistance through early activation of the host-defense genes in potato.Physiol Plant. 2009 May;136(1):19-29. doi: 10.1111/j.1399-3054.2009.01209.x. Epub 2008 Feb 4. Physiol Plant. 2009. PMID: 19508365
Cited by
-
Moving Beyond DNA Sequence to Improve Plant Stress Responses.Front Genet. 2022 Apr 19;13:874648. doi: 10.3389/fgene.2022.874648. eCollection 2022. Front Genet. 2022. PMID: 35518351 Free PMC article. Review.
-
Epigenetic regulation of plant immunity: from chromatin codes to plant disease resistance.aBIOTECH. 2023 Mar 17;4(2):124-139. doi: 10.1007/s42994-023-00101-z. eCollection 2023 Jun. aBIOTECH. 2023. PMID: 37581024 Free PMC article. Review.
-
Coordinated Epigenetic Regulation in Plants: A Potent Managerial Tool to Conquer Biotic Stress.Front Plant Sci. 2022 Jan 3;12:795274. doi: 10.3389/fpls.2021.795274. eCollection 2021. Front Plant Sci. 2022. PMID: 35046981 Free PMC article. Review.
-
Exploiting Epigenetic Variations for Crop Disease Resistance Improvement.Front Plant Sci. 2021 Jun 4;12:692328. doi: 10.3389/fpls.2021.692328. eCollection 2021. Front Plant Sci. 2021. PMID: 34149790 Free PMC article. Review.
-
Dynamics of H3K27me3 Modification on Plant Adaptation to Environmental Cues.Plants (Basel). 2021 Jun 8;10(6):1165. doi: 10.3390/plants10061165. Plants (Basel). 2021. PMID: 34201297 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

