The two-fold cost of sex: experimental evidence from a natural system
- PMID: 30233811
- PMCID: PMC6089407
- DOI: 10.1002/evl3.1
The two-fold cost of sex: experimental evidence from a natural system
Abstract
Over four decades ago, John Maynard Smith showed that a mutation causing asexual reproduction should rapidly spread in a dioecious sexual population. His reasoning was that the per-capita birth rate of an asexual population would exceed that of a sexual population, because asexual females do not invest in sons. Hence, there is a cost of sexual reproduction that Maynard Smith called the "cost of males." Assuming all else is otherwise equal among sexual and asexual females, the cost is expected to be two-fold in outcrossing populations with separate sexes and equal sex ratios. Maynard Smith's model led to one of the most interesting questions in evolutionary biology: why is there sex? There are, however, no direct estimates of the proposed cost of sex. Here, we measured the increase in frequency of asexual snails in natural, mixed population of sexual and asexual snails in large outdoor mesocosms. We then extended Maynard Smith's model to predict the change in frequency of asexuals for any cost of sex and for any initial frequency of asexuals. Consistent with the "all-else equal" assumption, we found that the increase in frequency of asexual snails closely matched that predicted under a two-fold cost.
Keywords: Potamopyrgus antipodarum; all-else-equal assumption; asexual reproduction; evolution of sex; experimental evolution; paradox of sex; sexual reproduction; two-fold cost of males.
Figures
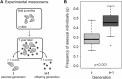
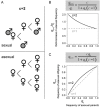

References
-
- Akaike, H. 1973. Information theory as an extension of the maximum likelihood principle Pp. 267–281 in Petrov B. and Csaki F., eds. Second international symposium on information theory. Akademiai Kiado, Budapest.
-
- Akaike, H. 1978. A Bayesian analysis of the minimum AIC procedure. Ann. Inst. Stat. Math. 30:9–14.
-
- Barŝiene, J. , Ribi G., and Barŝyte D.. 2000. Comparative karyological analysis of five species of Viviparus (Gastropoda: Prosobranchia. J. Moll. Stud. 66:259–271.
-
- Bell, G. 1982. The masterpiece of nature: The evolution and genetics of sexuality. California Univ. Press, Berkeley, CA.
-
- Bolker, B. 2008. Ecological models and data in R. Princeton Univ. Press, Princeton.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

