Osmolyte Induced Tumorigenesis and Metastasis: Interactions With Intrinsically Disordered Proteins
- PMID: 30234016
- PMCID: PMC6127622
- DOI: 10.3389/fonc.2018.00353
Osmolyte Induced Tumorigenesis and Metastasis: Interactions With Intrinsically Disordered Proteins
Abstract
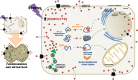
In spite of a great deal of work, the biochemical mechanisms underlying tumorigenesis and metastasis are not yet fully understood. Specifically regarding metastasis many authors consider that malignancy is caused by the accumulation of mutations. However, evidence is gathering to show that tumors are composed of heterogeneous cell populations subjected to selective pressures. In this micro evolutionary scenario, intra- and extra-cellular selective pressures will determine which subpopulations of tumor cells will thrive and be able to dissociate from the tumor as autonomous metastatic cells. We propose here that alteration of conformations of transcription factors confer novel non-canonical functions that may induce oncogenesis and metastasis in a mutation independent manner. We argue that the functional plasticity of transcription factors is due to intrinsically disordered domains (IDRs) of proteins. IDRs prevent spontaneous folding of proteins into well-defined three-dimensional structures. Because most transcription factors contain IDRs, each could potentially interact with many ligands. This high degree of functional pleiotropy would then be ultimately responsible for the metastatic phenotype. The conformations of proteins can be altered by chemical chaperones collectively known as osmolytes. Osmolytes are small organic molecules permeable through biological membranes that can accumulate in cells, increase the thermodynamic stability of proteins, modulate enzyme activity and prevent protein aggregation. Thus, by modifying IDRs, osmolytes could subvert the homeostatic regulatory network of cells. Untargeted metabolomic analysis of oral cancer cells showed that those with the greatest metastatic potential contained several osmolytes that were absent in the non-metastatic cells. We hypothesize that high concentrations of osmolytes might promote conformational alterations of transcription factors that favor metastatic behavior. This hypothesis is eminently testable by investigating whether: (a) the intracellular microenvironment of metastatic cells differs from non-metastatic cells and whether osmolytes are responsible for this change and (b) high intracellular concentrations of osmolytes are sufficient to induce structural modifications in regulatory protein so as to establish novel interactive networks that will constitute the metastatic phenotype. Synthetic cell penetrating peptides mimicking IDRs could act as sensitive probes. By exposing the peptides to the microenvironments of living tumor and metastatic tumor cells one should be able to compare the chemical shifts as revealed by spectra obtained by nuclear magnetic resonance (NMR).
Keywords: conformation; intrinsically disordered proteins; metastasis; osmolytes; transcription factors.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

