Pazopanib in patients with von Hippel-Lindau disease: a single-arm, single-centre, phase 2 trial
- PMID: 30236511
- PMCID: PMC6338228
- DOI: 10.1016/S1470-2045(18)30487-X
Pazopanib in patients with von Hippel-Lindau disease: a single-arm, single-centre, phase 2 trial
Abstract
Background: No approved systemic therapy exists for von Hippel-Lindau disease, an autosomal dominant disorder with pleiotropic organ manifestations that include clear cell renal cell carcinomas; retinal, cerebellar, and spinal haemangioblastomas; pheochromocytomas; pancreatic serous cystadenomas; and pancreatic neuroendocrine tumours. We aimed to assess the activity and safety of pazopanib in patients with von Hippel-Lindau disease.
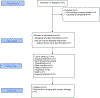
Methods: In this non-randomised, single-centre, open-label, phase 2 trial, adult patients with clinical manifestations of von Hippel-Lindau disease were recruited from the University of Texas MD Anderson Cancer Center (Houston, TX, USA) and were treated with pazopanib (800 mg orally daily) for 24 weeks, with an option to continue treatment if desired by the patient and treating physician. Primary endpoints were the proportion of patients who achieved an objective response and safety in the per-protocol population. The objective response was measured for each patient and each lesion type. Radiographic assessments were done at baseline and every 12 weeks throughout the study. Activity and safety were assessed with continuous monitoring and a Bayesian design. This study is registered with ClinicalTrials.gov, number NCT01436227, and is closed to accrual.
Findings: Between Jan 18, 2012, and Aug 10, 2016, we screened 37 patients with genetically confirmed or clinical features consistent with von Hippel-Lindau disease, of whom 31 eligible patients were treated with pazopanib. The proportion of patients who achieved an objective response was 42% (13 of 31 patients). By lesion sites responses were observed in 31 (52%) of 59 renal cell carcinomas, nine (53%) of 17 pancreatic lesions, and two (4%) of 49 CNS haemangioblastomas. Seven (23%) of 31 patients chose to stay on the treatment after 24 weeks. Four (13%) of 31 patients withdrew from the study because of grade 3 or 4 transaminitis, and three (10%) discontinued study treatment because of treatment intolerance with multiple intercurrent grade 1-2 toxicities. Treatment-related serious adverse events included one case each of appendicitis and gastritis and one patient had a fatal CNS bleed.
Interpretation: Pazopanib was associated with encouraging preliminary activity in von Hippel-Lindau disease, with a side-effect profile consistent with that seen in previous trials. Pazopanib could be considered as a treatment choice for patients with von Hippel-Lindau disease and growing lesions, or to reduce the size of unresectable lesions in these patients. The safety and activity of pazopanib in this setting warrants further investigation.
Funding: Novartis Inc and NIH National Cancer Institute core grant.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Figures

Comment in
-
The first prospective trial for von Hippel-Lindau disease: pazopanib.Lancet Oncol. 2018 Oct;19(10):1267-1269. doi: 10.1016/S1470-2045(18)30533-3. Epub 2018 Sep 17. Lancet Oncol. 2018. PMID: 30236512 No abstract available.
Similar articles
-
Belzutifan for patients with von Hippel-Lindau disease-associated CNS haemangioblastomas (LITESPARK-004): a multicentre, single-arm, phase 2 study.Lancet Oncol. 2024 Oct;25(10):1325-1336. doi: 10.1016/S1470-2045(24)00389-9. Epub 2024 Sep 13. Lancet Oncol. 2024. PMID: 39284337 Clinical Trial.
-
Variable response of CNS hemangioblastomas to Pazopanib in a single patient with von Hippel-Lindau disease: Case report.J Clin Neurosci. 2018 Apr;50:154-156. doi: 10.1016/j.jocn.2018.01.040. Epub 2018 Feb 1. J Clin Neurosci. 2018. PMID: 29396065
-
Repurposing propranolol as a drug for the treatment of retinal haemangioblastomas in von Hippel-Lindau disease.Orphanet J Rare Dis. 2017 Jun 29;12(1):122. doi: 10.1186/s13023-017-0664-7. Orphanet J Rare Dis. 2017. PMID: 28662711 Free PMC article.
-
Recurrent multiple CNS hemangioblastomas with VHL disease treated with pazopanib: a case report and literature review.CNS Oncol. 2015;4(6):387-92. doi: 10.2217/cns.15.22. Epub 2015 Oct 26. CNS Oncol. 2015. PMID: 26497655 Free PMC article. Review.
-
Pancreatic neuroendocrine tumours in patients with von Hippel-Lindau disease.Endokrynol Pol. 2020;71(3):256-259. doi: 10.5603/EP.a2020.0027. Endokrynol Pol. 2020. PMID: 32797472 Review.
Cited by
-
The β2-adrenergic receptor antagonist ICI-118,551 blocks the constitutively activated HIF signalling in hemangioblastomas from von Hippel-Lindau disease.Sci Rep. 2019 Jul 11;9(1):10062. doi: 10.1038/s41598-019-46448-6. Sci Rep. 2019. PMID: 31296894 Free PMC article.
-
Cancer Genetics and Therapeutic Opportunities in Urologic Practice.Cancers (Basel). 2020 Mar 18;12(3):710. doi: 10.3390/cancers12030710. Cancers (Basel). 2020. PMID: 32197306 Free PMC article. Review.
-
Predominantly Solid Hemangioblastoma Presenting as an Extra-Axial Cerebellopontine Angle Lesion.Cureus. 2021 Feb 2;13(2):e13071. doi: 10.7759/cureus.13071. Cureus. 2021. PMID: 33680613 Free PMC article.
-
Von Hippel-Lindau Disease: Current Challenges and Future Prospects.Onco Targets Ther. 2020 Jun 16;13:5669-5690. doi: 10.2147/OTT.S190753. eCollection 2020. Onco Targets Ther. 2020. PMID: 32606780 Free PMC article. Review.
-
Understanding the Impact of Belzutifan on Treatment Strategies for Patients with VHL.J Kidney Cancer VHL. 2022 Sep 28;9(3):41-46. doi: 10.15586/jkcvhl.v9i3.245. eCollection 2022. J Kidney Cancer VHL. 2022. PMID: 36310638 Free PMC article.
References
-
- Ho TH, Jonasch E. Genetic kidney cancer syndromes. Journal of the National Comprehensive Cancer Network : JNCCN. 2014;12(9):1347–55. - PubMed
-
- Latif F, Tory K, Gnarra J, Yao M, Duh FM, Orcutt ML, et al. Identification of the von Hippel-Lindau disease tumor suppressor gene. Science. 1993;260(5112):131720. - PubMed
-
- Maxwell PH, Wiesener MS, Chang GW, Clifford SC, Vaux EC, Cockman ME, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399(6733):271–5. - PubMed
-
- Lonser RR, Glenn GM, Walther M, Chew EY, Libutti SK, Linehan WM, et al. von Hippel-Lindau disease. Lancet. 2003;361(9374):2059–67. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

