Nucleosides isolated from Ophiocordyceps sinensis inhibit cigarette smoke extract-induced inflammation via the SIRT1-nuclear factor-κB/p65 pathway in RAW264.7 macrophages and in COPD mice
- PMID: 30237706
- PMCID: PMC6136406
- DOI: 10.2147/COPD.S172579
Nucleosides isolated from Ophiocordyceps sinensis inhibit cigarette smoke extract-induced inflammation via the SIRT1-nuclear factor-κB/p65 pathway in RAW264.7 macrophages and in COPD mice
Abstract
Background: Ophiocordyceps sinensis (C. sinensis) extracts have been found to have a therapeutic effect on patients with chronic obstructive pulmonary disease (COPD). Silent information regulator 1 (SIRT1) plays an important role in the regulation of inflammatory mediators and correlates with lung function and COPD exacerbations. The objective of this work was to explore the anti-inflammatory effect and preliminary pathways of nucleosides from cultured C. sinensis on RAW264.7 macrophages and COPD mice.
Materials and methods: The nucleosides were extracted from cultured C. sinensis powder and further purified by macroporous resin D101 and glucan G10 columns. Inflammation and oxidative stress models in RAW264.7 macrophages and in mice were established by injection of cigarette smoke extract (CSE). We then examined how the isolated nucleosides regulated the production of the associated inflammatory mediators in vitro and in vivo by enzyme-linked immunosorbent assay, reverse transcription polymerase chain reaction, and Western blot.
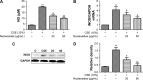
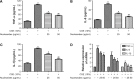
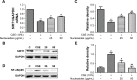
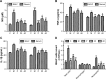
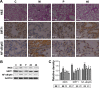
Results: The nucleosides inhibited inflammatory mediator expression of tumor necrosis factor-α, interleukin-6, interleukin-1β, and nitric oxide in both the CSE-stimulated RAW264.7 macrophages and mice. Moreover, the nucleosides elevated SIRT1 activation and suppressed nuclear factor-κB (NF-κB)/p65 activation in vitro and in vivo. Nucleoside treatment significantly decreased the levels of the inflammatory mediators in the bronchoalveolar lavage fluid (BALF) and serum of the CSE-induced mice. The nucleosides also altered the recruitment of inflammatory cells in BALF and improved characteristic features of the lungs in the CSE-induced mice.
Conclusion: These results show that the nucleosides suppressed COPD inflammation through the SIRT1-NF-κB/p65 pathway, suggesting that the nucleosides may be partly responsible for the therapeutic effects of cultured C. sinensis on COPD patients.
Keywords: COPD; NF-κB/p65; Ophiocordyceps sinensis; SIRT1; anti-inflammatory; nucleosides.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures



Similar articles
-
Ergosterol attenuates cigarette smoke extract-induced COPD by modulating inflammation, oxidative stress and apoptosis in vitro and in vivo.Clin Sci (Lond). 2019 Jul 15;133(13):1523-1536. doi: 10.1042/CS20190331. Print 2019 Jul 15. Clin Sci (Lond). 2019. PMID: 31270147
-
Sirtuin regulates cigarette smoke-induced proinflammatory mediator release via RelA/p65 NF-kappaB in macrophages in vitro and in rat lungs in vivo: implications for chronic inflammation and aging.Am J Physiol Lung Cell Mol Physiol. 2007 Feb;292(2):L567-76. doi: 10.1152/ajplung.00308.2006. Epub 2006 Oct 13. Am J Physiol Lung Cell Mol Physiol. 2007. PMID: 17041012
-
Bone marrow mesenchymal stem cells ameliorate lung injury through anti-inflammatory and antibacterial effect in COPD mice.J Huazhong Univ Sci Technolog Med Sci. 2017 Aug;37(4):496-504. doi: 10.1007/s11596-017-1763-3. Epub 2017 Aug 8. J Huazhong Univ Sci Technolog Med Sci. 2017. PMID: 28786060
-
Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders.Cell Signal. 2013 Oct;25(10):1939-48. doi: 10.1016/j.cellsig.2013.06.007. Epub 2013 Jun 11. Cell Signal. 2013. PMID: 23770291 Review.
-
Myricetin inhibits TNF-α-induced inflammation in A549 cells via the SIRT1/NF-κB pathway.Pulm Pharmacol Ther. 2020 Dec;65:102000. doi: 10.1016/j.pupt.2021.102000. Epub 2021 Feb 16. Pulm Pharmacol Ther. 2020. PMID: 33601000 Review.
Cited by
-
K63 Ubiquitination of P21 Can Facilitate Pellino-1 in the Context of Chronic Obstructive Pulmonary Disease and Lung Cellular Senescence.Cells. 2022 Oct 3;11(19):3115. doi: 10.3390/cells11193115. Cells. 2022. PMID: 36231077 Free PMC article.
-
scRNA-seq and scATAC-seq analyses highlight the role of TNF signaling pathway in chronic obstructive pulmonary disease model mice.PLoS One. 2025 May 9;20(5):e0322538. doi: 10.1371/journal.pone.0322538. eCollection 2025. PLoS One. 2025. PMID: 40343927 Free PMC article.
-
Attenuation of Cigarette-Smoke-Induced Oxidative Stress, Senescence, and Inflammation by Berberine-Loaded Liquid Crystalline Nanoparticles: In Vitro Study in 16HBE and RAW264.7 Cells.Antioxidants (Basel). 2022 Apr 28;11(5):873. doi: 10.3390/antiox11050873. Antioxidants (Basel). 2022. PMID: 35624737 Free PMC article.
-
Circ-RBMS1 Knockdown Alleviates CSE-Induced Apoptosis, Inflammation and Oxidative Stress via Up-Regulating FBXO11 Through miR-197-3p in 16HBE Cells.Int J Chron Obstruct Pulmon Dis. 2021 Jul 16;16:2105-2118. doi: 10.2147/COPD.S311222. eCollection 2021. Int J Chron Obstruct Pulmon Dis. 2021. PMID: 34295155 Free PMC article.
-
Accumulation Characteristics of Natural Ophiocordyceps sinensis Metabolites Driven by Environmental Factors.Metabolites. 2024 Jul 27;14(8):414. doi: 10.3390/metabo14080414. Metabolites. 2024. PMID: 39195510 Free PMC article.
References
-
- Trifilieff A, Ethell BT, Sykes DA, et al. Comparing the cardiovascular therapeutic indices of glycopyrronium and tiotropium in an integrated rat pharmacokinetic, pharmacodynamic and safety model. Toxicol Appl Pharmacol. 2015;287(1):9–16. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

